 |
 |
- Search
| Phys Act Nutr > Volume 25(4); 2021 > Article |
|
Abstract
[Purpose]
In this study, we investigated whether a 70% ethanolic (EtOH) extract of Sargassum horneri had antioxidant and anti-inflammatory effects in lipopolysaccharide (LPS)-stimulated macrophage-like RAW 264.7 cells.
[Methods]
The proximate composition, fatty acids, amino acids, and dietary fiber of S. horneri, various biologically active compounds, and antioxidant activity were analyzed.
[Results]
The DPPH and ABTS free radical scavenging activities, as well as the reduction power, of the S. horneri extract used here were significantly increased in a concentration-dependent manner. This indicates that S. horneri contains bioactive compounds, such as phenols and flavonoids, that have excellent antioxidant activity. The cellular viability and metabolic activity results confirmed that the extract had no discernible toxicity at concentrations up to 100 μg/mL. The levels of nitrites and cytokines (PGE2, TNF-α and IL-6), which mediate pro-inflammatory effect, were significantly inhibited by treatment with either 50 or 100 μg/mL S. horneri extract, whereas that of IL-1β was significantly inhibited by treatment with 100 μg/mL of the extract. Similarly, the expression of iNOS and COX-2 proteins also decreased according to 50 or 100 μg/mL extract concentrations. NF-κB binding to DNA was also significantly inhibited by treatment with 100 μg/mL of extract.
Oxidation products are produced when a substance loses hydrogen or electrons upon chemical reaction with oxygen. This process thought to cause aging or cancer and is related to the inhibition of DNA synthesis [1]. Inflammation, which is one of the biological responses to noxious stimuli, is a mechanism that restores damaged cells while removing the noxious entity [2]. However, a persistent inflammatory response can lead to cell necrosis and disease [3]. The inflammatory proteins iNOS and COX-2, and mediators of inflammation such as NO and cytokine, induce various inflammatory diseases, affect tumor formation, and further affect morbidity and mortality [4].
The production of pro-inflammatory cytokines, such as PGE2, TNF-α, IL-1β and IL-6, increases during strenuous and prolonged exercise [5-7]. The production of prostaglandin E2 (PGE2) is part of the inflammatory response triggered by micro-trauma in skeletal muscles [8,9]. In addition, the overt signs and symptoms of inflamed tissue include swelling, redness, pain, and reduced function [9]. Recently, various physiological effects, such as anti-inflammatory, antioxidant, and antihypertensive activities have been confirmed, attracting attention not only in food research but also in terms of potential pharmaceutical applications [10]. Sargassum horneri is a species of brown algae, rich in bioactive substances. This yellowish-brown algal species has a cylindrical stem and long oval-shaped leaves and is mainly distributed along the coast of Korea, Japan, and China [11]. In Korea, it is considered as waste stemming from large-scale inflow and is mainly used as feed. S. horneri has undergone extensive study, including analyzing the content of its functional polysaccharide fucoidan [12], its potential as a functional ingredient for whitening and wrinkle improvement [13], prevention of osteoporosis [14], and antioxidant and anti-inflammatory activity [15] while additional uses of functional materials in various fields are expected to be reported in future studies [16].
In this study, the contents of general components, fatty acids, constituent amino acids, free amino acids, and dietary fiber were analyzed in dried raw powder of S. horneri. The antioxidant activity of S. horneri ethanolic extract was evaluated by analyzing the 2,2-Diphenyl-1-picryldrynzyl (DPPH) and 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical scavenging ability and reducing power, as well as the total phenols and flavonoids content. To assess the anti-inflammatory activity of S. horneri, RAW 264.7 macrophage cells were treated with the dried ethanol extract, and the production of nitrite and related cytokines (PGE2 - Prostaglandin E2, TNF-α - Tumour necrosis factor, IL-6 - Interleukin-6 and IL-1β - Interleukin-1β), the expression of inflammatory proteins (iNOS - inducible nitric oxide synthase, COX-2 - cyclooxygenase-2) and nuclear factor kappa B (NF-κB) DNA binding activity were also investigated.
S. horneri was provided by Jeju National University (Jeju city, South Korea). The raw material dried using sea breeze was used to analyze the general components, fatty acids, amino acids, and dietary fiber. The ethanolic S. horneri extract was generated by adding 10 g of dried S. horneri to 500 mL of 70% EtOH, and the extraction was repeated three times while stirring at room temperature for 12 h. The obtained extract was concentrated under reduced pressure, freeze-dried, and then used to analyze antioxidant and anti-inflammatory activities.
The proximate composition and dietary fiber content were analyzed using dried extraction according to the Association of Official Analytical Chemists (AOAC) method [17]. Moisture quantification was determined by atmospheric drying, ash content was analyzed by sintering at 550 ℃, crude protein content was determined using the Kjeldahl method, and crude fat analyzed following the Soxhlet method. The carbohydrate content was calculated using Equation (1).
The insoluble dietary fiber content was analyzed using an enzymatic-gravimetric method. Dried S. horneri (0.5 g) was added to 40 mL of MES-TRIS buffer, mixed, and reacted with α-amylase solution and protease enzyme solution. This was followed by the addition of 5 mL of 0.561N HCl, adjust the pH to 4.0-4.7, and the sample was then filtered. The residual part was washed sequentially with distilled water, ethanol (95%), and acetone, after which the content was determined, and the crude protein and crude content were measured and subtracted to determine the insoluble dietary fiber content. The quantitative soluble dietary fiber content was analyzed using liquid chromatography. The insoluble dietary fiber content and high-molecular-weight/low-molecular-weight ratio were calculated, and the soluble dietary fiber content was calculated by subtracting the insoluble dietary fiber content. The total dietary fiber content was calculated by adding the values for the insoluble and the soluble dietary fiber contents.
For the analysis of fatty acids, 2 g of S. horneri dried sample was used for extraction using chloroform-methanol, and 100 mg of concentrated lipid was collected after filtering under reduced pressure, then mixed with 3 mL of 1N KOH-ethanol with stirring, and 3 mL of 14% BF3-methanol was added, followed by heating at 80 °C for 5 min to form methyl ester. Subsequently, 3mL of saturated NaCl solution and 1.5mL of hexane were added to a test tube to collect the upper aliquot. Anhydrous Na2SO4 was added to remove moisture, and the sample was analyzed using gas chromatography (GC) (Shimadzu GC-17A, Shimadzu Co., Tokyo, Japan), which was fused with a silica capillary column (SP™-2560, 100 m × 0.25 mm ID, 0.25 μm film thickness, Bellefonte, PA, USA). To perform the fatty acid analysis, we used an isothermal period for 5 min at 100 °C. The temperature was increased by 3.5 °C/min up to 240 °C, and the isothermal period was maintained for 30 min. Subsequently, 2 μL of the sample was injected into the column. The injector and detector ports were maintained at 260 °C using helium gas. The results for the total fatty acid content are expressed as g/100 g of sample, and the results for the individual fatty acids are expressed as weight percentage (%) of the total fatty acids.
A 0.02 g sample of dried S. horneri, was mixed with 15 mL of 6N HCl and the container was filled with N2 gas, sealed under reduced pressure, and the sample was hydrolyzed at 110 °C for 24 h in a dry oven. The hydrolyzed sample was acid-removed with a vacuum concentrator at 50 °C using a dilution buffer (pH 2.2). After filtration using a 0.2 μm membrane, the sample was analyzed using an automatic amino acid analyzer (S433-H, SYKAM, Gewerbering 15, Germany).
To analyze the free amino acid content, 1 g of dried S. horneri was extracted by adding distilled water (20 mL) to collect the supernatant. The supernatant was purified with 10 mL of 0.12N lithium citrate buffer (pH 2.2) using a rotary evaporator to remove the distilled water. A 0.2 g quantity of sulfosalicylic acid was added to the fixed solution and allowed to stand at 4 °C for 1 h. Then, it was filtered through a 0.2 μm membrane filter, diluted, and analyzed using an automatic amino acid analyzer (S430, SYKAM). The analysis conditions for both the compositional and the free amino acids using a cation separation column (LCA K07/Li), size (4.6 × 150 mm), were at 37~74 ℃ temperatures, a buffer flow rate of 0.45 mL/min, a reagent flow rate of 0.25 mL/min, pH 3.45 to 10.85 for the compositional amino acids and pH 2.9 to 7.95 for the free amino acids, with measurement at wavelengths of 440 nm and 570 nm using a fluorescence spectrophotometer.
To evaluate the antioxidant activity using the DPPH and ABTS radical scavenging assays, the dried 70% EtOH extract was diluted to different concentrations, and 0.2 mM DPPH was added at a 1:1 ratio, followed by reaction at room temperature for 30 min, and the absorbance was measured at 517 nm. For the ABTS radical, 7.4 mM ABTS was prepared using PBS buffer, 2.6 mM potassium persulfate was prepared and mixed 1:1, and the mixture was then kept in a dark place at room temperature for one day. When ABTS and PBS were mixed at a ratio of 1:15, the absorbance at 734 nm was 0.7. The S. horneri 70% EtOH extract was diluted to different concentrations, the reagent and the sample were mixed at a ratio of 950:50 and reacted in a dark room for 30 min at room temperature. The absorbance was then measured at 734 nm. The DPPH and ABTS radical scavenging ability were calculated using Equation (2).
The S. horneri 70% EtOH extract was diluted to different concentrations, and 100 μL of 0.2 M sodium phosphate buffer (pH 6.6) was added to 100 μL of the sample. After adding, 100 μL of 10% potassium ferricyanide, the sample was allowed to react at 50 °C for 20 min. After adding 100 μL (100 mg/mL) of 10% trichloroacetic acid, centrifugation was performed at 1000 × g for 10 min. The supernatant was mixed with 0.1% ferric chloride (1 mg/mL) at a ratio of 5:1, and the absorbance was measured at 700 nm.
The phenol content analysis was conducted according to the Folin-Denis [18] method. The S. horneri 70% EtOH extract was diluted to concentrations, and 1 mL of Folin-Ciocalteu reagent and 10% Na2CO3 solution were added to 1 mL of the sample and reacted at room temperature for 1 h. The absorbance was then measured at 700 nm. The total phenol content was calculated using a calibration curve obtained with gallic acid as a standard material. To analyze the flavonoid content according to the method of Jia [19], the 70% EtOH extract of S. horneri was diluted to different concentrations, and 30 μL of 5% NaNO2 was added to 1 mL of the sample and allowed to react for 5 min. After adding 30 μL of 10% AlCl3 and 200 μL of 1M NaOH, the absorbance was measured at 510 nm. Qercetin was used as a standard, and a calibration curve was obtained to calculate the flavonoid content.
We used mouse-derived macrophage RAW 264.7 cell line, which was purchased from the Korea Cell Line Bank (Seoul, Korea). Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% heat-inactivated FBS, streptomycin (100 mg/mL), penicillin G (100 units/mL), and L-glutamine (2 mM) were used to culture the cells in an incubator at 37 °C and 5% CO2.
Cell viability was analyzed as described by Berridge and Tan [20]. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, a tetrazole (MTT) (50 mg/mL) was added to each well containing cells (1 × 105 cell/mL) for 4 h. The formed formazan was dissolved in dimethyl sulfoxide (DMSO), and the absorbance was measured at 540 nm.
Nitrite formation was measured according to the Griess reagent reaction method [21]. In this process, 100 μL of Griess reagent (0.1% (w/v) N-(1-naphathyl)-ethylenediamine and 1% (w/v) sulfanilamide in 5% (v/v) phosphoric acid) was added to 100 μL of each supernatant and thoroughly mixed for 10 min. The absorbance was then measured at 540 nm.
RAW 264.7 cells (1 × 105 cell/mL in 48-well plates) were treated with S. horneri extract at different concentrations and 1 μg/mL lipopolysaccharide (LPS) was added to each concentration for 24 h. The final sample was centrifuged at 13,000 × g for 2 min, and the supernatant was collected and analyzed using an enzyme-linked immunosorbent kit from R&D Systems, Inc. (Minneapolis, MN, USA) to measure PGE2, TNF-α, IL-1β, and IL-6 levels following the manufacturer’s protocol.
iNOS, COX-2, and NF-κB protein expression levels were analyzed using western blotting. The cells were dissolved in protease inhibitor mix and 20 mM Tris-HCl buffer. Protein concentrations were measured using Bio-Rad protein assay dye reagent concentrate (#5000006, Bio-Rad Laboratories, Hercules, CA, USA). After electrophoresis process on a 12% sodium dodecyl sulfate-polyacrylamide gel, the proteins were transferred to a nitrocellulose membrane (Bio-Rad Laboratories). The membranes were blocked with 5% skim milk and then incubated with primary antibodies (Santa Cruz Biotechnology, Texas, USA) followed by horseradish peroxidase (HRP)-conjugated secondary antibodies. The protein bands were detected using the light emitted by ECL reagent (Amersham Pharmacia Biotech, Piscataway, NJ, USA) [22].
To obtain cytoplasmic and nuclear fractions, an extraction Kit (Cayman, Ann Arbor, MI, USA) was used following the manufacturer’s instruction. To analyze the DNA-binding activity of NF-κB, the extracted nuclear fraction was used and an NF-kB transcription factor assay kit (Cayman) was used according to the manufacturer’s protocol.
The results were obtained by repeating the experiment three times, and the data are presented as means ± standard deviation (SD). Statistically significant differences were considered at p <0.05, using one-way analysis of variance (ANOVA) Tukey’s multiple comparison test with GraphPad Prism software (Version 5.01, Graphpad Software, Inc., San Diego, CA, USA).
The proximate analysis and dietary fiber content indicated that the level of moisture was 14.2%, carbohydrates 46%, ash 21%, crude fat 4.4%, and crude protein approximately 15% in S. horneri dried powder, as shown in Table 1. The total content of dietary fiber was 55.3%, and the content of insoluble dietary fiber was 51.2%, accounting for most of the total dietary fiber. The soluble dietary fiber content was 4.1%, which was lower than that of the insoluble dietary fiber.
The fatty acid composition results indicated that the total saturated fatty acid content was 69.26%, with a palmitic acid (C16:0) content of 9.63%, heptadecanoic acid (C17:0) 20.53% and tricosanoic acid (C23:0) at 39.1%. The total monounsaturated fatty acids (approximately 3.26%) included 1.44% palmitoleic acid (C16:1) and 1.82% oleic acid (C18:1n9c). The total polyunsaturated fatty acid content was 27.48%, comprising cis-11,14-Eicosadienoic acid (C20:2) (1.85%) and cis-5,8,11,14,17-Eicosapentaenoic acid (C20:5n3) (25.63%) in S. horneri dried powder, as shown in Table 2.
The S. horneri dried powder compositional amino acid analysis results (Table 3) indicate that the total 16 compositional amino acids content was 8,574.29 mg/100 g. Of these, the glutamic acid (1,203.49 mg/100 g) and aspartic acid (970.297 mg/100 g) contents were the highest, while the histidine (254.255 mg/100 g), methionine (235.754 mg/100 g), and tyrosine (182.62 mg/100 g) contents were the lowest. The remaining compositional amino acids such as threonine, serine, proline, glycine, alanine, valine, isoleucine, leucine, phenylalanine, lysine, and arginine were present at approximately 448-746 mg/100 g.
The free amino acid content assay detected seven different free amino acids, and the quantified amount was 51.68 mg/100 g, as shown in Table 4. Of these, the glutamic acid content was the highest, at 25.056 mg/100 g, followed by alanine at 8.59 mg/100 g, phosphoserine at 7.221 mg/100 g, and aspartic acid at 6.019 mg/100 g. These constituted more than 90% of the total free amino acids and are considered to be the main free amino acids.
The antioxidant activity of the S. horneri extract, as determined using DPPH and ABTS radical scavenging assays, is presented in Figure 1. The DPPH radical scavenging activity significantly increased in a concentration-dependent manner from 0.0625 mg/mL to 4 mg/mL of extract, as shown in Figure 1(A). Similarly, the ABTS radical scavenging activity also significantly increased in a concentration-dependent manner, as shown in Figure 1(B). Moreover, the scavenging ability at 1 mg/mL of S. horneri extract revealed a radical scavenging ability of 40.1% for DPPH and 36.9% for ABTS.
A reducing power experiment was conducted to investigate the antioxidant activity of the 70% EtOH extract of S. horneri (Figure 2). The results indicated that the extract at concentrations from 0.5 mg/mL to 4 mg/mL significantly increased the reducing power. At 1 mg/mL of extract, the absorbance was 0.20.
The analysis of S. horneri ethanol extract showed a total phenol content of approximately 8.52 mg GAE/g and a total flavonoid content of 82.17 mg QE/g (Table 5), indicating that bioactive compounds, specifically flavonoids, with antioxidant activity are present at significant levels.
To investigate the effect of the 70% EtOH extract of S. horneri on RAW 264.7 cells, the cytotoxicity of the extract was evaluated using the MTT assay. The results indicated that the S. horneri extract had no discernible cytotoxicity at concentrations up to 100 μg/mL (Figure 3). Therefore, the subsequent experiments were carried out at a maximum concentration of 100 μg/mL.
The nitrite assay was performed as an indicator of intracellular NO production. Nitrite levels were analyzed in LPS-induced cells after treatment with S. horneri extract at concentrations of 25‒100 μg/mL. The results in Figure 4 indicate that nitrite production gradually decreased with increasing extract concentrations compared with control LPS-induced cells.
When the S. horneri 70% EtOH extract was used at different concentrations, the production of PGE2, TNF-α, and IL-6 was significantly reduced in the 50 and 100 μg/mL treatment groups compared to the LPS-treated cells, as shown in Figure 5(A‒D). IL-1β levels were significantly reduced in the 100 μg/mL treatment group.
The protein expression results showed that the expression of iNOS and COX-2 was increased in LPS-induced cells (Figure 6), whereas when the cells were treated with S. horneri extract, the expression of the pro-inflammatory proteins iNOS and COX-2 decreased in a concentration-dependent manner.
The DNA binding activity of NF-κB (Figure 7) was increased by LPS, whereas there was a significant concentration-dependent decrease in its DNA-binding activity in the S. horneri extract-treated groups compared to the LPS induced group.
This study analyzed the proximate components, such as moisture, ash, carbohydrates, crude protein, and crude fat in dried S. horneri. A similar proximate composition was identified by Boo et al. [23] in S. horneri collected in April. Brown algae generally contain a large proportion of carbohydrates, including polysaccharides such as laminarin and fucoidan, which are physiologically active substances [24]. In addition, the mineral content related to physiological control functions was reported to be higher than that of land plants [25]. However, the results differ slightly from those of Kim et al. [26], which is thought to be due to the differences in the collection location and time of the same type of seaweed [27]. The dietary fiber in seaweed impedes the excretion of heavy metals and salt in the human body and prevents cholesterol from depositing on the inner walls of the blood vessels [28]. The current study indicated that S. horneri has a high percentage (55.3%) of total dietary fiber content.
The fatty acid analysis results indicated that the saturated tricosanoic (C23:0) fatty acid and polyunsaturated cis-5,8,11,14,17-eicosapentaenoic (C20:5n3) fatty acid, which belong to the ω-3 series, accounted for 64.73% of the total. Polyunsaturated fatty acids are essential fatty acids that cannot be produced in the body and must be obtained directly by the consumption of food [29]. Polyunsaturated fatty acids of the ω-3 series, which are present at low levels in land organisms but are known to be abundant in seafood, have physiological activity and help prevent heart and vascular diseases [30]. Hong et al. [31] reported that polyunsaturated fatty acids are present at approximately 27.4% in marine algae of Miyeok (Undaria pinnatifida) and fatty acids are present at 24.7% in Pare (Enteromorpha compressa) algae. The current study suggests that S. horneri algae have similar percentages of polyunsaturated fatty acids.
The compositional amino acid analysis results showed that glutamic acid, aspartic acid, and leucine accounted for approximately 34% of the total amino acid content, and these are considered to be the main constituent amino acids. Glutamic and aspartic acids have been reported to be present at higher levels than most amino acids in seaweed, accounting for 22‒44% of the amino acids in brown algae [32]. The free amino acid analysis revealed that glutamic acid and alanine were the most abundant, accounting for 65% of the total free amino acids.
The antioxidant activity of S. horneri was investigated based on the ability to reduce DPPH. The DPPH radical has a relatively stable structure, and this type of reactive oxygen species is thought to promote aging [33]. This assay method confirmed that the 70% S. horneri extract had a concentration-dependent free radical scavenging ability. In addition, the ABTS radical scavenging activity, which can evaluate both hydrophilic and hydrophobic samples, and the scavenging ability of free radicals induced by reaction with potassium persulfate, were also assayed [34]. As was the case with DPPH, the ABTS radical scavenging ability was significantly increased according to the concentration of the extract. Shin et al. [35] reported that the ethanol extract of brown algae has both DPPH and ABTS radical scavenging ability at increasing concentrations. In terms of reducing radical power, the absorbance value showed a tendency to increase as the concentration of the extract increased. Cho et al. [36] reported a similar trend, with the antioxidant activity of the ethanol extract of seaweed increasing according to the concentration. Phenols and flavonoids exhibit bioactive effects, such as antioxidant and antibacterial activities [37]. In this study, the total levels of phenols and flavonoids were found to be 8.52 mg GAE/g and 82.17 mg QE/g, respectively. The total flavonoid level was relatively high based on the results of an analysis of ten domestic brown algae [38]. Phenolic compounds have a phenolic hydroxyl group that can bind to macromolecules, such as proteins, and possess physiological activity and excellent free radical scavenging ability. Although synthetic antioxidants such as BHT and BHA are widely used, the importance of developing natural antioxidants is emerging due to their efficiency [39,40]. The 70% ethanol extract of S. horneri exhibited significant antioxidant activity and has ample potential for use as a natural antioxidant.
To examine the anti-inflammatory activity of S. horneri, cell viability was first analyzed using the MTT assay, and it was confirmed that there was no discernible toxicity up to an extract concentration of 100 μg/mL. LPS treatment induced inflammation in the macrophage RAW 264.7 cell line. The nitrites generated under inflammatory conditions were inhibited upon treatment with the S. horneri extract in a concentration-dependent manner. The PGE2, TNF-α, and IL-6 analysis results indicated that the group treated with extracts at concentrations of 50 and 100 μg/mL showed significant inhibitory effects, and for IL-1β this occurred at a concentration of 100 μg/mL. These inflammatory cytokines trigger nitrite production [41]. The inflammatory protein iNOS produces NO, whereas COX-2 produces PGE2 [42]. In this study, the expression of these inflammatory proteins decreased according to the extract concentration. Moreover, the DNA-binding activity of NF-κB, which regulates inflammatory mediators, was significantly inhibited upon treatment with the extract. When NF-κB is activated, it induces the production of inflammatory cytokines and proteins [43]. Therefore, these results suggest that S. horneri is a functional food that may improve performance during repeated engagement in high-intensity exercise. Moreover, it may maintain muscle integrity by reducing muscle damage and inflammatory responses as a result of its ability to attenuate the production of iNOS, COX-2, PGE2, and NO. Similar results were observed with creatine supplementation, which after five days of treatment resulted in reduced levels of pro-inflammatory cytokines after 24 to 48 h of high-intensity exercise [44].
In conclusion, the results of this study indicate that S. horneri contains various components that have beneficial physiological activities and that can prevent vascular diseases. In addition, it was possible to confirm the excellent antioxidant and anti-inflammatory activities of 70% EtOH S. horneri extract. Therefore, S. horneri has ample potential for use in the field of natural medicines and in functional foods for athletes.
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1D1A1B01006822).
Figure 1.
(A) DPPH and (B) ABTS radicals scavenging activity (%) of S. horneri 70% EtOH extract (mg/mL). All values are mean ± SD.
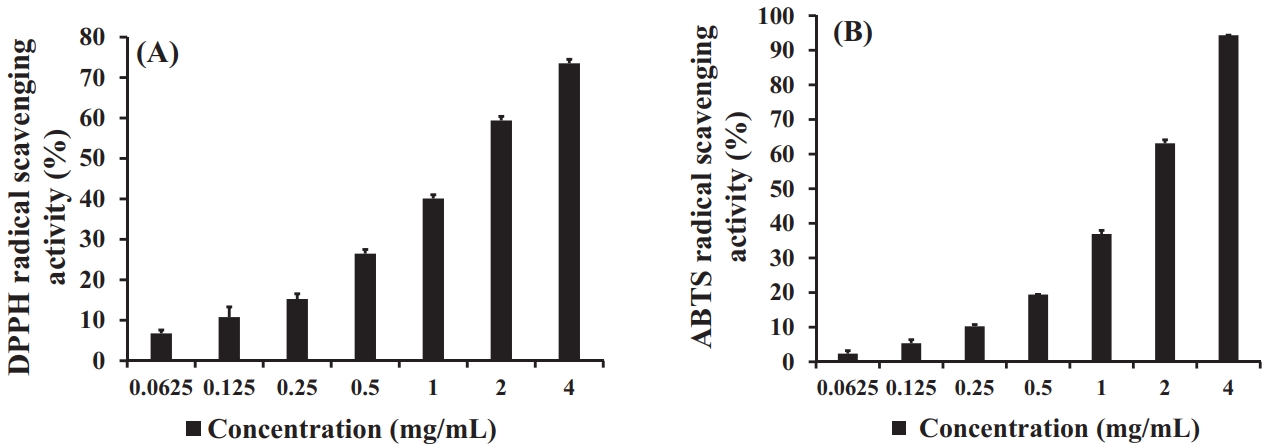
Figure 3.
Effect of S. horneri 70% EtOH extract (μg/mL) on RAW 264.7 cell viability. All values are mean ± SD.
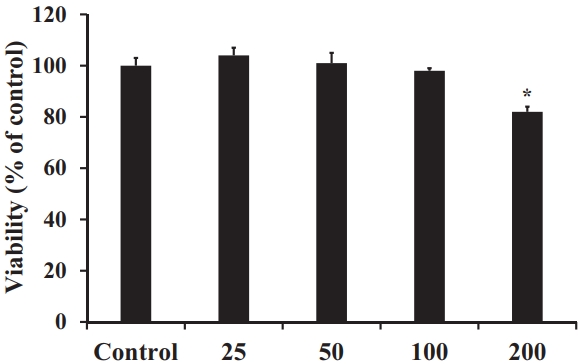
Figure 4.
Effect of S. horneri 70% EtOH extract (μg/mL) on nitrite production in RAW 264.7 cells. All values are mean ± SD.
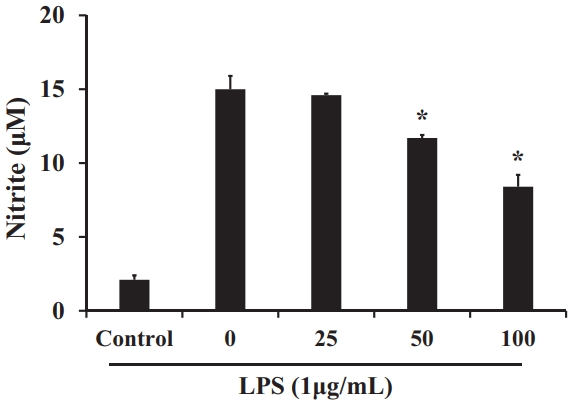
Figure 5.
Effect of S. horneri 70% EtOH extract (μg/mL) on the production of (A) PGE2, (B) TNF-α, (C) IL-1β, and (D) IL-6 in RAW 264.7 cells. All values are mean ± SD.
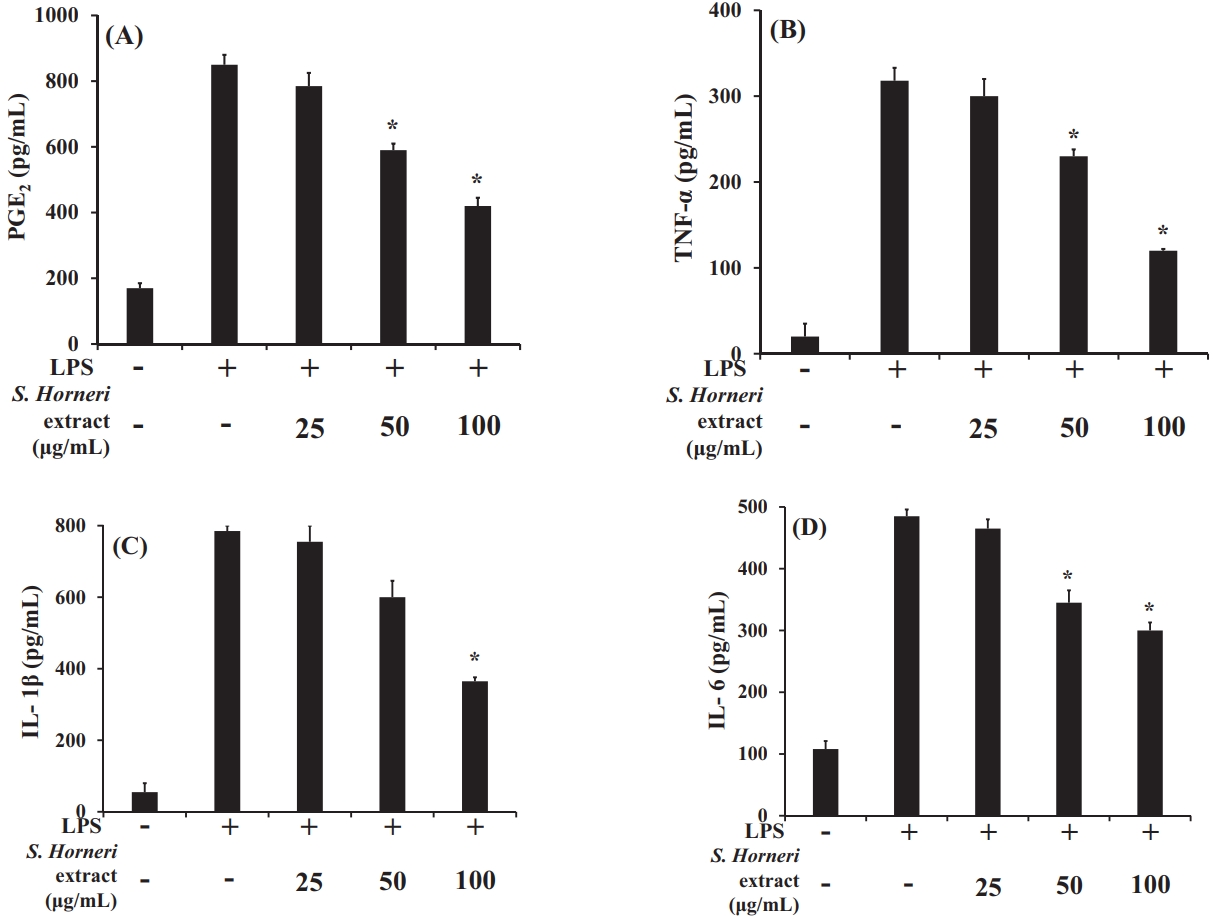
Figure 6.
Effect of S. horneri 70% EtOH extract (μg/mL) on expression of iNOS and COX-2 in RAW 267 cells.
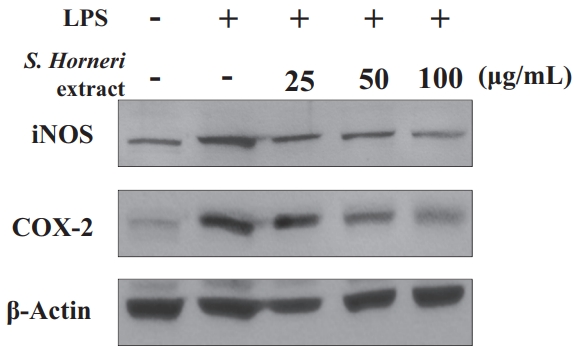
Figure 7.
Effect of S. horneri 70% EtOH (μg/mL) extract on expression on DNA binding activity of NF-kB in RAW 267 cells. All values are mean ± SD.
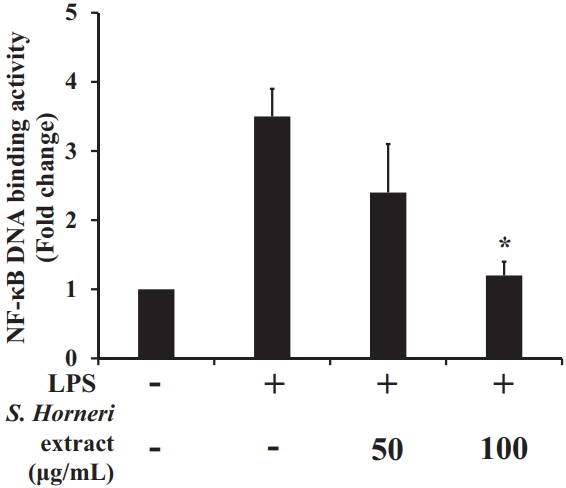
Table 1.
Proximate composition and dietary fiber contents of S. horneri dried sample.
| Proximate composition (% dry wt.) | Dietary fiber contents (%) | ||||||
|---|---|---|---|---|---|---|---|
| Moisture | Carbohydrates | Ash | Crude fat | Crude protein | IDF1) | SDF2) | TDF3) |
| 14.2 ± 0.4 | 46 ±1.6 | 21 ± 0.4 | 4.4 ± 0.1 | 15 ± 1.1 | 51.2 | 4.1 | 55.3 |
Table 2.
Composition of fatty acids (%) in S. horneri.
Table 3.
Compositional amino acids contents of S. horneri dried powder.
REFERENCES
1. Kim HK, Kim YE, Do JR, Lee YC, Lee BY. Antioxidative activity and physiological activity of some korean medicinal plants. Korean J Food Sci Technol 1995;27:80-5.
2. Baek YM, Choi JY, Lee CW, Jeon YS, Han JT, Jang SI, Yoo HS. Effects of chinemys reevesii on lipopolysaccaride-indused inflammatory reactions. J Physiol & Pathol Korean Med 2012;26:26-34.
3. Yu AR, Park HY, Kim YS, Ha SK, Hong HD, Choi HD. Immuno-enhancing effect of seed extracts on a RAW 264.7 macrophage cell line. J Korean Soc Food Sci Nutr 2012;41:1671-6.

4. Kim JA, Ha SE, Kim SM, Vetrivel P, Kim GS, Kim EH. Anti-inflammatory activities of sinensetin on LPS-stimulated L6 skeletal muscle cells. J Biomed Transl 2019;20:1-7.

5. Malm C. Exercise-induced muscle damage and inflammation: fact or fiction? Acta Physiol Scand 2001;171:233-9.


6. Clarkson PM, Hubal MJ. Exercise-induced muscle damage in humans. Am J Phys Med Rehabil 2002;81:S52-S69.


7. Nosaka K, Newton M. Concentric or eccentric training effect on eccentric exercise-induced muscle damage. Med Sci Sports Exerc 2002;31:63-9.

8. Bansil CK, Wilson GD, Stone MH. Role of prostaglandins E and F2a in exercise-induced delayed muscle soreness. Med Sci Sports Exerc 1985;17:276-80.
9. Bassit RA, Curi R, Costa Rosa LFBP. Creatine supplementation reduces plasma levels of pro-inflammatory cytokines and PGE2 after a half-ironman competition. Amino Acids 2008;35:425-31.


10. Lee MS, Lee YK. Effects of seaweeds consumption on obesity and food intake. J of The Korea Contents Association 2014;14:478-85.
11. Eom GH, Shin JB, Lim SJ, Lee KT, Han YS, Lee KJ. Evaluation of dietary supplementation of Sargassum horneri for pacific white shrimp litopenaeus vannamei. Korean J Fish Aquat Sci 2020;53:909-17.
12. Hong JH, Lee MJ, Moon SJ, Kim DS, Nho EY, Choi JY, Kim KS. Analysis of monosaccharide composition of fucoidan from Sargassum horneri using UPLC-DAD-TOF/MS combined with precolumn derivatization by 3-methyl-1-phenyl-5-pyrazolone. Korean J Food Preserv 2019;26:219-27.

13. Gam DH, Hong JW, Jeon SJ, Baek DH, Kim JW. Development of ultrasound-assisted extraction for production of bioactive compounds with whitening and anti-wrinkle effects from Sargassum horneri. KSBB J 2020;35:294-302.

14. Yamaguchi M. Marine alga Sargassum horneri component and bone homeostasis: role in osteoporosis prevention. Int J Food Sci Nutr 2013;2:9-14.
15. Shao P, Chen X, Sun P. Chemical characterization, antioxidant and antitumor activity of sulfated polysaccharide from Sargassum horneri. Carbohydr. Polym 2014;105:260-9.
16. Park SR, Thomas SS, Cha YS, Kim KA. Inhibitory effects of Sargassum horneri extract against endoplasmic reticulum stress in HepG2 cells. J Nutr Health 2020;53:583-95.

17. AOAC Official methods of analysis, 15th ed. Association of Official Analytical Chemists. Washington, DC, USA. 1990;788.
18. Folin O, Denis W. On phosphotungstic-phosphomolybdic compounds as color reagents. J Biol Chem 1912;12:239-49.

19. Jia Z, Tang M, Wu J. The determination of flavonoid contents in mulberry and their scavenging effects on super-oxide radicals. Food Chem 1999;64:555-9.

20. Berridge MV, Tan AS. Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyl tetrazolium bromide (MTT): subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Arch Biochem Biophys 1993;303:474-82.


21. Titheradge MA. The enzymatic measurement of nitrate and nitrite. Methods Mol Biol 1998;100:83-91.


22. Ko W, Sohn JH, Jang JH, Ahn JS, Kang DG, Lee HS, Kim JS, Kim YC, Oh H. Inhibitory effects of alternaramide on inflammatory mediator expression through TLR4-MyD88-mediated inhibition of NFκB and MAPK pathway signaling in lipopolysaccharide-stimulated RAW264.7 and BV2 cells. Chem Biol Interact 2016;244:16-26.


23. Boo HJ, Moon HS, Jee YH, Jeon YJ, Chun JY. Appearance characteristics and antioxidant activity of Jeju coast Sargassum horneri according to collecting time. J Korean Soc Food Sci 2020;49:485-92.

24. Ko SC, Kang SM, Ahn G, Yang HP, Kim KN, Jeon YJ. Antioxidant activity of enzymatic extracts from Sargassum coreanum. J Korean Soc Food Sci Nutr 2010;39:494-9.

25. Lee JM, You SG, Kim SM. Functional activities of low molecular weight peptides purified from enzymatic hydrolysates of seaweeds. J Korean Soc Food Sci 2005;34:1124-29.

26. Kim HS, Asanka Sanjeewa KK, Shanura Fernando IP, Ryu BM, Yang HW, Ahn G, Kang MC, Heo SJ, Je JG, Jeon YJ. A comparative study of Sargassum horneri Korea and China strains collected along the coast of Jeju island South Korea: its components and bioactive properties. Algae 2018;33:341-9.

27. Cho DM, Kim DS, Lee DS, Kim HR, Pyeun JH. Trace components and functional saccharides in Seaweed-1-Changes in proximate composition and trace elements according to the harvest season and places. J Korean Fish Soc 1995;28:49-59.
28. Kim ET, Oh GH. Effect of glutathione with sea tangle extract on prevention of selenite-induced cataract formation in rats. J Korean Ophthalmol Soc 2009;50:1555-62.

29. Seo UH, Kang HJ, Yoon KB, An YJ, Kim JB. Analysis of dietary fiber, mineral content and fatty acid composition in cheonggak (codium fragile). Korean J Food & Nutr 2019;32:328-34.
30. Kang JY, Park NH, Hai LQ, Seo MS, Hong YK. Comparison of aquacultured and wild undaria pinnatifida thalli for evaluation of eight quality standards. Korean J Fish Aquat Sci 2008;41:337-42.

31. Hong JS, Kwon YJ, Kim YH, Kim MK, Park IW, Kang KH. Fatty acid composition of miyeok (undaria pinnatifida) and pare (enteromorpha compressa). J Korean Soc Food Sci Nutr 1991;20:376-80.
32. Fleurence J. Seaweed proteins: biochemical, nutritional aspects and potential uses. Trends Food Sci Technol 1999;10:25-8.
33. Kim HS, Im NR, Park JH, Kim MO, Park SN. Antioxidative effect and active component analysis of gnaphalium affine D. Don. extraxts. J Soc Cosmet Sci Korea 2014;40:11-20.

34. Kim MK. Comparison of antioxidant and antimicrobial activities of pulp and peel extracts of mango. J Invest Cosmetol 2017;13:113-8.

35. Shin SH, Kang SH. The antioxidation effect of brown algae extract. J Korean Soc Cosmetol 2021;27:851-8.

36. Cho ML, Yoon SJ, Kim YB. The nutritional composition and antioxidant activity from undariopsis peterseniana. Ocean Polar Res 2013;35:273-80.

37. Baek JY, Lim SY. Flavonoid and phenol contents and antioxidant effect of wine by-product extracts. J Life Sci 2016;26:948-54.

38. Lee CH, Park YN, Lee SG. Analysis and comparison of bioactive compounds and total antioxidant capabilities of Korean brown algae. Korean J Food Sci Technol 2020;52:54-9.
39. Kim YJ. Evaluation of antioxidant activity and thermal stability of plant polyphenols. Biomater Res 2009;13:30-6.
40. Choi SY, Cho HS, Sung NJ. The antioxidative and nitrite scavenging ability of solvent extracts from wild grape (vitis coignetiea) skin. J Korean Soc Food Sci Nutr 2006;35:961-6.

41. Seo SH, Lee H, Choi MO. The anti-inflammatory actions and dermal bioactive effects of coreopsis lanceolata extracts. J Korean Soc Cosmetol 2018;24:472-81.
42. Lee DS, Kim KS, Li B, Choi HG, Keo S, Jun KY, Park JH, Kim YC. Anti-inflammatory effect of the cirsium japonicum var. ussuriense 70% ethanolic extract in RAW 264.7 cells by heme oxygenase-1 expression. Saengyak Hakhoe Chi (ISO4) 2012;43:39-45.
- TOOLS
-
METRICS

-
- 2 Crossref
- Scopus
- 2,880 View
- 46 Download
- Related articles in Phys Act Nutr






