Effects of individualized low-intensity mat Pilates on aerobic capacity and recovery ability in adults
Article information
Abstract
[Purpose]
Although Pilates is one of the most widely performed physical activities in Korea, no physiological evidence is available regarding its energy recovery ability. Therefore, the purpose of this study was to investigate the effects of individualized low-intensity mat Pilates on aerobic capacity and recovery ability in adults.
[Methods]
Ten physically active women participated in this study. Pre- and post-lactate threshold (LT) tests were performed to compare jogging/running speeds (S; km·h−1) and heart rates (HR; beats·min−1) at 1.5, 2.0, 3.0, 4.0 mmol·L−1 lactate concentrations (La−). Subjects performed 1 h of low-intensity mat Pilates twice a week for four weeks. During these sessions, exercise intensity was determined based on the heart rate corresponding to individualized low-inten- sity recovery zone 1, which was estimated using a mathematical model of log-log LT1 (from pre-test; < 2 mmol·L−1). All physiological variables were measured before and after exercise intervention.
[Results]
Significant differences were found in body mass increase and body mass index increase between the pre- and post-tests (p = 0.016 and p = 0.014, respectively, effect size (ES) = 0.13; ES = −0.11). Levels of La− between 1.0 and 1.4 m·s−1 in the post-LT test tended to decrease, although such decrease was not significantly different. Moderate to high positive correlations between differences (Δ) of S and ΔHR at 1.5, 3.0, and 4.0 mmol·L−1La− were observed.
[Conclusion]
Positive correlations between ΔS and ΔHR at certain La− levels indicate that low-intensity mat Pilates based on heart rate corresponding to individualized recovery zone 1 might be recommended for physically active adults.
INTRODUCTION
Mitochondrial dysfunction and oxidative stress are involved in aging, age-related diseases, and metabolic syndromes. Metabolic syndrome is a cluster of pathological conditions, including abdominal obesity, hyperlipidemia, insulin resistance, and other abnormalities [1,2]. This new non-communicable disease has become apparent as a common public health problem globally [2,3]. Increased risk of developing metabolic syndrome is seen in the general population in the modern workplace, mostly due to a sedentary lifestyle, high-calorie intake, low-fiber food consumption, and technological development [2-4]. Individuals with metabolic syndrome are more likely to develop cardiovascular diseases and type 2 diabetes than those without metabolic syndrome [5]. The cost of curing metabolic syndrome is a social, governmental, and economic burden worldwide [3,5-8].
Therefore, many health organizations have recommended aerobic endurance exercises to prevent risk factors for this syndrome 8 . Physical activity such as aerobic endurance training is recommended to regulate metabolic risk factors, including fasting triglycerides, high-density lipoprotein (HDL), and insulin sensitivity, and to improve cardiorespiratory fitness and type 2 diabetes mellitus [4,9]. However, the volume and intensity of physical activity differ among studies, with most of these activities achieving minimal amount of physical activity [10,11]. Exercising moderately for at least 150 minutes or vigorously for 75 minutes per week was suggested by the World Health Organization [11]. In this regard, individually prescribed exercise improves physical health in the general population [12]. Lactate threshold (LT) tests have been used by scientists and clinical physicians for several decades as a useful physiological metric to measure aerobic endurance capability and prescribe individualized exercise intensity for athletes as well as cardiac patients [12-14]. Therefore, blood lactate concentration is recognized as a more sensitive physiological parameter than estimated VO2max or maximal heart rate (HRmax) [13,15].
Blood lactate level during LT test is used to individualize exercise intensity for training program [12,14,16]. Exercise intensity areas based on the LT test included training zones (1, 2, and 3: low, threshold/moderate, and high) [12,13]. Blood lactate values increase with increasing exercise intensity because more pyruvate and lactate are generated through carbohydrate metabolism [12,13,17]. Previous studies have suggested that measured blood lactate concentration and fat and carbohydrate metabolism are indirect variables of metabolic flexibility and mitochondrial function [12,13,18]. Low-intensity training (< 2 mmol·L-1) can increase mitochondrial function, such as metabolic flexibility [12-14]. Triglycerides in fat cells are hydrolyzed to glycerol and free fatty acids (FFAs), which are converted into acetyl-CoA by β-oxidation in the mitochondria [13,17,19]. For obese individuals or the general population or both, low-intensity exercise (LIE) might be preferable to high-intensity exercise (HIE) training. Before participating in HIE, building an aerobic base is first required through high-volume and low-intensity training based on the typical linear periodized program to reduce stress on skeletal muscles, lower the risk of injuries, and increase adherence to the training [10,12,20]. Currently, Pilates as an alternative physical exercise program is widely performed by the general population [21]. Pilates can improve muscular strength, core stability, flexibility, dynamic posture maintenance, and balance [21,22]. According to a survey conducted by the Ministry of Culture, Sports, and Tourism in 2019 on national recreational sports trends, Pilates is listed as one of the top ten physical activities of Koreans [23]. Despite the popularity of Pilates exercises, little is known about more accurate and individualized exercise intensity to ensure better health and performance.
Previous studies have indicated that participation in Pilates exercises is accompanied by health benefits [21,22]. It is currently unclear how individualized low-intensity mat Pilates exercises impact aerobic and endurance capacity in adults because there are fewer studies regarding the relationship of low-intensity exercise based on the LT test with improvement in physiological parameters, particularly mat-based Pilates exercise. A mat Pilates exercise program was selected to make the exercise intervention easier for participants without space and time constraints. Moreover, mat Pilates was effective enough to modify a 1 h long sequence, which was confirmed in our pilot study. Therefore, this study aimed to investigate the effects of individualized low-intensity mat Pilates exercise on energetic recovery ability (ATP re-synthesis from accumulated blood lactate) and general endurance in adults using several physiological parameters, including the LT test.
METHODS
Participants
The sample size was calculated as follows: effect size = 1.20, alpha error probability = 0.05, statistical power = 0.80 (G* power software version 3.1.9.7; Franz Faul, University Kiel, Germany) [12,18,24]. A total of ten physically active female adults participated in this study (age: 31 ± 5 years; height: 160 ± 5 cm; body mass: 55.1 ± 6.5 kg; body mass index: 21 ± 2.8 kg·m−2; Table 1). Participants participated in group exercise classes for at least two to three sessions per week for at least one year. They were recruited from regional fitness centers and Pilates studios through official notifications. Participants were asked to avoid intake of food or drinks except for water for at least three hours before each testing session to avoid the influence on measurements due to food intake [12,25,26]. All participants completed anthropometric measurements using a bioelectrical impedance analyzer (Inbody 270; Inbody Co. Ltd., Seoul, Korea) before and after the exercise intervention (Table 1). Pre- and post-lactate threshold (LT) tests were performed to compare speeds (S; km·h-1) and heart rates (HR; beats·min-1) at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 lactate concentrations (La−) using a mathematical interpolation model [12,27-29]. Participants performed low-intensity mat Pilates based on an individually prescribed HR range under the guidance of an instructor. The exercise intervention was performed for 1 h twice a week for four weeks [12]. This study was approved by the Institutional Ethics Committee of the CHA University (No. 1044308-202111-HR-087-02). The approved protocols were per the Declaration of Helsinki.
Study design
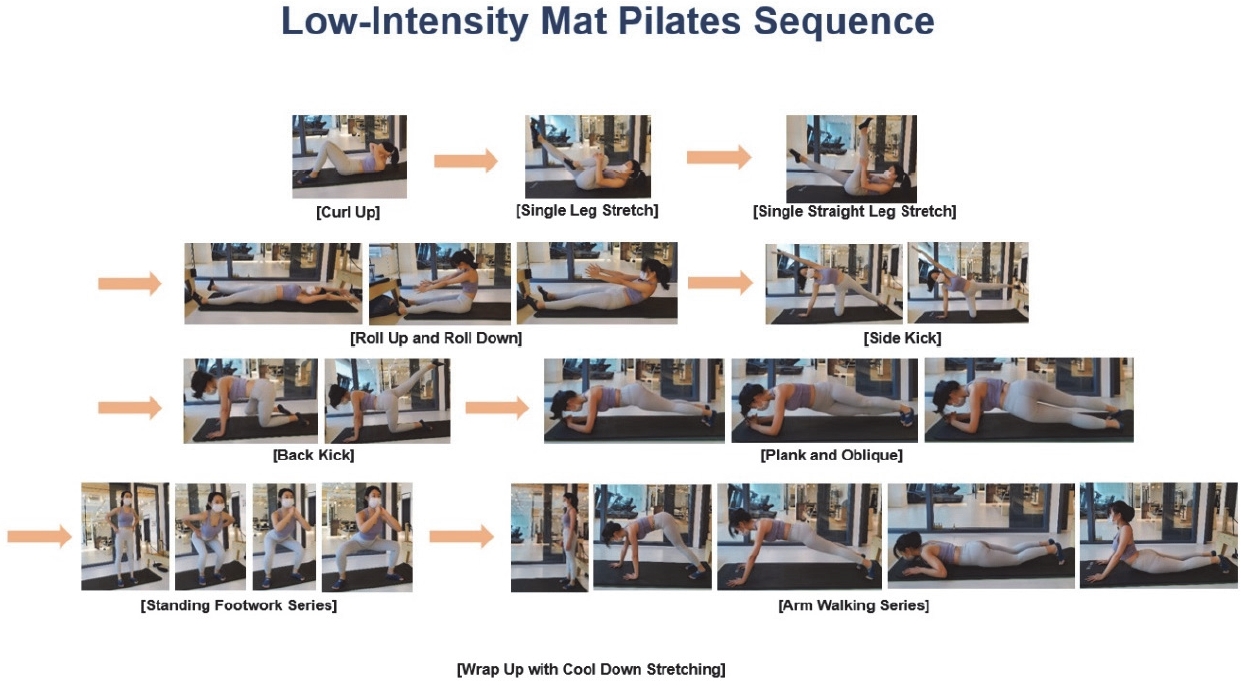
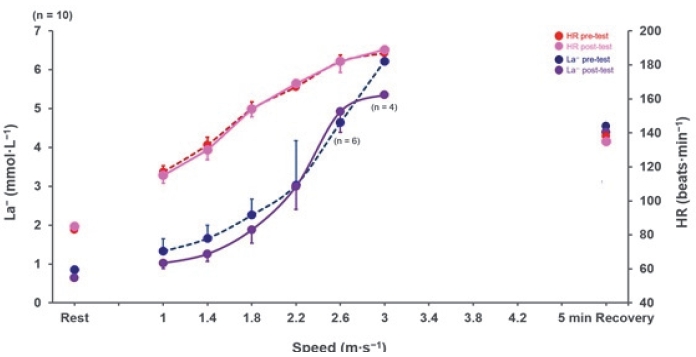
All participants completed an anthropometric measurement and LT test on a treadmill (NR30XA, DRAX Corporation Ltd., Seoul, Korea) before and after the exercise intervention. The pre- and post-LT tests consisted of 5 min stages interspersed with 30 s breaks for La− measurements. The first stage was started at 1.0 m·s−1, with increments of 0.4 m·s−1 every 5 minutes [25,27,30]. All the procedures were conducted in a controlled laboratory environment (room temperature, 23 °C; relative humidity, 50%). Capillary blood sampling was performed from the earlobe (20 μL) to determine La− levels. The blood lactate concentration was analyzed using an enzymatic-amperometric sensor chip system (Biosen C-line, EKF diagnostics sales, GmbH, Barleben, Germany). The test was ceased when La− exceeded 4.0 mmol·L−1 after each exercise test in all participants [12,25,31]. Following the test, every participant went through the recovery stage for 5 min at the same speed as the first stage to check the tendency of individual decreased lactate concentrations (Figure 1). Based on pre-LT test results, participants performed an hour of individualized low-intensity mat Pilates two times a week for four weeks to achieve the same exercise volume. A four-week exercise intervention period was selected to maintain the same low-intensity exercise volume as in previous studies [12,26,32,33]. The individualized low-intensity heart rate for the exercise intervention was calculated from the results of the pre-LT test in 72% of LT 1 (101 ± 17 beats·min−1) heart rate within recovery zone 1 (recovery domain before the increased lactate curve within zone 1; RZ 1) [14,25,34] (Table 2). The workout intensity was based on the heart rate corresponding to 72% of LT 1 (< 2 mmol·L −1 blood lactate) from the results of the pre-LT test. LT 1 was estimated using a previously described mathematical model of log-log LT 1 [12,25,27-29,35]. The mat Pilates sequence for exercise intervention was modified from that used in previous studies 36-38 (Figure 2). During exercise intervention, the HR of all participants was carefully managed within the individualized HR range using an HR telemetry (H10 sensor, Polar Electro, Finland) [12,39].

Study design.
The pre-LT test was conducted before the 4-week low-intensity (< 2 mmol∙L-1) mat Pilates exercise intervention. The LT test consisted of 5 min stages interspersed with 30 s breaks for La− measurements. The first stage was started at 1.0 m·s−1, with increments of 0.4 m·s−1 every 5 min. Capillary blood sampling was performed from the earlobe (20 μL) to determine La−. During the intervention, two sessions of 1 h mat Pilates were performed every week for four weeks. After the 4-week exercise intervention, a post-LT test was conducted for all participants.
Statistical analyses
All data were statistically analyzed using GraphPad Prism 9.0 (GraphPad Software, La Holla, CA, USA). The parameters are presented as the mean and standard deviation (mean ± SD) or standard error of the mean (S.E.M). Normal distribution was assessed using the Shapiro–Wilk test. A paired t-test was used to compare pre- and post-anthropometric data and La− and HR variables between the pre- and post-LT tests. A Wilcoxon signed-rank test was used to compare pre- and post-test body fat, exercise speeds at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 La−, and HR at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 La−. The effect sizes (ES; Cohen’s d or Z/√N) were calculated for the parameters. The thresholds for small, moderate, and large effects were 0.2, 0.5, and 0.8, respectively, for the parametric tests and 0.1, 0.3, and 0.5, respectively, for non-parametric tests [40]. Statistical significance was set at p < 0.05. Furthermore, Spearman’s rank correlations between ΔS and ΔHR at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 La− were analyzed.
RESULTS
Comparison of pre- and post-test anthropometric data
Significant differences were found in body mass increase and body mass index increase after the individualized low-intensity mat Pilates exercise intervention compared with those before the exercise intervention (p = 0.016 and p = 0.014, respectively, effect size (ES) = 0.13; ES = −0.11). The body fat percentage showed no significant difference between the pre-test and post-test (p > 0.05).
Comparison of pre- and post-LT test S and HR at La−
There was no significant difference in HR values at 1.5, 2.0, 3.0, or 4.0 mmol·L−1 La− between before and after exercise intervention (p > 0.05). Levels of La− between 1.0 and 1.4 m·s-1 in the lactate threshold test after exercise intervention tended to decrease in zone 1. However, there was no significant difference in exercise speed at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 La− between the pre- and post-LT test (p > 0.05) (Figure 3).
Correlations between ∆S and ∆HR of all subjects at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 La−
Moderate to high positive correlations between ∆S and ∆HR at 1.5 (correlation coefficient [r] = 0.70; R2 = 0.49; 95% confidence interval [95% CI]: 24.13–53.73; p = 0.0252), 3.0 (r = 0.83; R2 = 0.68; 95% CI: 37.58–68.61; p = 0.0043), and 4.0 mmol·L-1 La− (r = 0.84; R2 = 0.70; 95% CI: 26.68–78.88; p = 0.0034) were observed.
DISCUSSION
The physiological effects of Pilates exercises, especially on aerobic and recovery abilities, are currently unclear. To the best of our knowledge, this study is the first to examine how individualized low-intensity mat Pilates influence aerobic endurance and energetic recovery ability in female adults. The principal finding of this study was that body mass and body mass index values significantly increased after the exercise intervention. Levels of La− between 1.0 and 1.4 m·s-1 in the lactate threshold test after exercise intervention tended to decrease in zone 1. Furthermore, moderate to high positive correlations were observed between ∆S and ∆HR for all subjects at 1.5, 3.0, and 4.0 mmol·L-1 La− after the 4-week individualized low-intensity mat Pilates exercise intervention.
Previous studies have shown that low-load-high repetitions training sessions can result in body fat reduction [21,41]. A 10-week program of a 1-hour mat Pilates session showed significant body fat reduction in both male and female participants over the exercise intervention period [38]. A low-load-high repetition exercise intervention for 12 weeks significantly reduced the body fat percentage [41]. However, no changes in body fat were found after low-intensity Pilates exercise in this study. This indicates that a higher number of repetitions and a longer intervention period might improve body composition and reduce body fat in participants [42].
Levels of La− between 1.0 and 1.4 m·s-1 after exercise intervention showed a tendency of improvement, whereas no significant difference was observed in HR values or exercise speed at 1.5, 2.0, 3.0, or 4.0 mmol·L−1 La− (Figure 3). Previous studies have shown that endurance training could increase total fat utilization while decreasing the use of carbohydrates [43-46]. Therefore, the tendency of a rightward shift in the exponential lactate curve between 1.0 and 1.4 m·s-1 La− was understandable. During resting and low-intensity exercise conditions, more plasma fatty acids are delivered to produce energy, and the rate of lactate production is lowered [13,17,19]. In the post-LT test, the levels of La− in the last stage showed a tendency to improve, while there were no significant differences between stages. Our findings suggest that low-intensity mat Pilates exercise for four weeks might improve general endurance. Lee et al. [12] and Hwang et al. [25] reported that LIE and individualized low-intensity exercise (ILIE) improved energy recovery and general endurance in zones 1, 2, and 3. There was no significant difference in the HR values after four weeks of 1 h of low-intensity Pilates exercise. Previous studies on LIE have analyzed several HR-related variables, such as heart rate variability (HRV), HRmax , and blood pressure [47-49]. A recent study showed that HRV in patients with peripheral artery disease improved after five sessions of exercise intervention per week for 3 months [48]. Moreover, 3-5 sessions of 1 h LIE for 12 weeks led to increased left ventricular ejection and decreased blood pressure in LIE for at least 12 weeks, which might improve the HR variables. Furthermore, Hwang et al. [25] reported that the 9-week ILIE within recovery zone 1 (RZ 1) decreased the HR in professional athletes.
Although there were no significant differences in HR values or calculated jogging/running speeds at 1.5, 2.0, 3.0, and 4.0 mmol·L−1 La− in the pre- and post-tests, correlation analyses of the study presented positive relationships between ∆S and ∆HR at 1.5, 3.0, and 4.0 mmol·L-1 La− (Figure 4). A previous study by Lee et al. [12] has reported that after four weeks of 1 h LIE intervention, HR levels of physically active adults tend to increase with improved exercise speeds, showing positive, moderate to high correlations between ∆S and ∆HR at 1.5, 2.0, and 4.0 mmol·L−1 La−. Hwang et al. [25] have reported that after nine weeks of 1 h ILIE sessions within RZ 1 for professional athletes, La− levels and HRs are lowered in the post-LT test with low positive correlations between ∆S and ∆HR at 1.5 and 2.0 mmol·L−1 La−. These studies indicate that exercise intervention based on heart rate corresponding to recovery training zone 1 from the results of the LT-test may be prescribed to physically active adults, but not for professional athletes, since only low positive correlations between ∆S and ∆HR were observed in professional soccer players after a 9-week of 1-h ILIE (72% of log-log LT) [25].
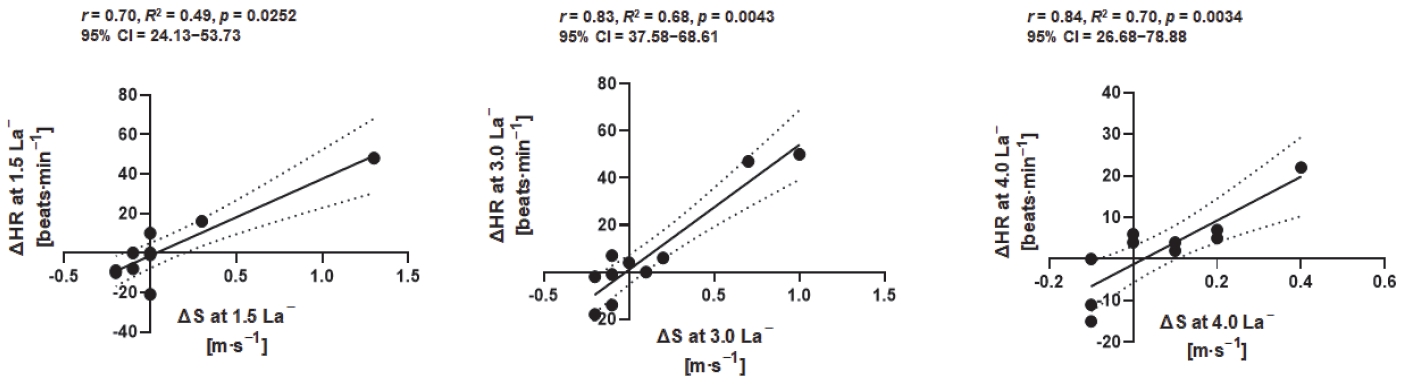
Spearman’s correlation (n = 10) between delta (∆) exercise speeds and delta (∆) heart rate at 1.5 (r = 0.70, p = 0.0252, 95% CI: 24.13–53.73), 3.0 (r = 0.68, p = 0.0043, 95% CI: 37.58–68.61), and 4.0 mmol·L−1 La− (r = 0.84, p = 0.0034, 95% CI: 26.68–78.88) showed moderate to high positive relationships.
Pilates exercises may significantly benefit cardiopulmonary physiology [38]. During resting and LIE conditions, lactate produced in active muscle cells is transported to the liver or kidney (Cori cycle) via the blood and resynthesized by the cell-to-cell lactate shuttle mechanism, also known as gluconeogenesis [12,13,50]. The movement pattern of the upper and lower limbs collectively allows a higher oxygenated blood flow into muscle tissues, enhancing muscle-oxidative capacity [37,51,52]. Since lactate metabolism is closely related to hepatic blood flow, increased local circulation in muscles during low-intensity Pilates exercises may improve cardiorespiratory fitness [13,37]. According to previous studies, forearm isometric strength is inversely related to blood lactate concentrations, especially in smaller muscle groups, such as those in the forearms and muscle groups that do not have much impact on total lactate production [24,50,53,54]. During prolonged exercise lasting for two hours, the elimination of lactate mostly occurs and is maintained in the liver [13]. Splanchnic uptake of lactate is higher after arm exercises compared with that after leg exercises [24,50,55]. Therefore, arm exercise has a greater effect on the hepatic uptake of lactate than leg exercise [50,56,57]. The increased hepatic uptake of lactate during arm movements affects ATP re-synthesis in the liver [50,58,59]. Therefore, more arm-based movements should be included in the latter part of the low-intensity mat Pilates sequence to improve aerobic endurance and energy recovery ability.
Previous studies have shown that exercise sessions with a higher training volume could increase indices of metabolic cost and aerobic fitness [56,60]. Arterial lactate concentration decreases during prolonged exercise owing to lowered lactate release from the working muscles [13,61]. According to Tinoco-Fernández et al. [38], 10-weeks of three 1-h mat Pilates sessions each week significantly improved cardiorespiratory variables, such as peak V̇O2, maximal V̇O2, HR, and respiratory exchange ratio (RER). Adults who have not been physically active or practiced exercise habitually showed substantial cardiorespiratory adaptation after the Pilates exercise intervention [22,38]. In contrast, no significant cardiorespiratory adaptation was observed in a 4-week Pilates program [62]. Therefore, to improve cardiovascular health and aerobic performance with the Pilates method, exercise intervention may be required to be performed for a longer time.
For further studies, calorie and nutritional intake control of participants should be conducted before the experimental protocol. It is necessary to instruct participants not to change their diet habits during the experimental period [41,42,52]. Moreover, no control group was included in this study. This limitation should be addressed in future studies.
Our findings indicate that individualized low-intensity Pilates exercises may enhance aerobic endurance and energy recovery in adults. The tendency of a rightward shift of the exponential lactate curve can be indirectly explained by improved energetic recovery ability, including fat oxidation and ATP re-synthesis, along with aerobic endurance capacity in zones 1, 2, and 3 [12,13,25,31]. If low-intensity Pilates exercise is performed for more than 6-8 weeks, due to its low energy consumption, Pilates exercise would improve the mechanism of fat metabolism and mitochondrial function [12,13,25,31]. Future studies with larger sample sizes are required to investigate how individualized low-intensity mat Pilates is related to other metabolism-related indices such as triglyceride, HDL-cholesterol, and fasting plasma glucose.
According to the findings of this study, low-intensity mat Pilates based on heart rate corresponding to 72% of LT 1 may be prescribed to physically active adults. LIE based on heart rate may be suitable for exercise beginners and physically active individuals but not for professional athletes. Along with low-intensity mat Pilates, aerobic exercise activities, such as low-intensity jogging/running, are recommended to improve cardiovascular fitness and mitochondrial function, resulting in improved metabolic flexibility and general endurance. Traditionally, Pilates exercises have concentrated on flexibility, body control, muscle strength, and accuracy of movements. However, the physiological parameters found important in this study are useful for people who perform Pilates exercises. Our findings indicate that individualized low-intensity Pilates exercises may enhance aerobic endurance and energy recovery in adults. The tendency of a rightward shift of the exponential lactate curve can be indirectly explained by improved energetic recovery ability, including fat oxidation and ATP re-synthesis, as well as aerobic endurance in zones 1, 2, and 3. If low-intensity Pilates exercise is performed for more than 6-8 weeks, due to its low energy consumption, Pilates exercise would improve the mechanism of fat metabolism and mitochondrial function. To achieve physical health benefits and cardiorespiratory fitness with mat Pilates, it is recommended that ILIE in RZ 1 be performed with multiple sets, a high number of repetitions, and a short interval, which can then enable the general population to exercise in higher volumes in zones 2 and 3.
Acknowledgements
We would like to thank all the participants for their involvement in this study.