INTRODUCTION
Atopic dermatitis (AD) is a major chronic or relapsing inflammatory disease of the skin that often leads to asthma and other allergic disorders [1]. It is accompanied by pruritic and eczematous skin lesions [2]. Recently, the incidence of AD has rapidly increased and has become a major public health concern worldwide [3]. Although the pathogenesis of AD is not entirely understood, genetic susceptibility, the host’s environment, skin barrier dysfunction, bacterial infection, and immunological factors appear to play a role in its development. The acute phase of AD is strictly related to increased T helper 2 (Th2) lymphocytes and basophils, while T helper 1 (Th1) cell-mediated AD lesions are found in chronic AD [2,4]. Furthermore, activated mast cells, increased immunoglobulin E (IgE), and increased levels of inflammatory cytokines are also found in AD skin lesions [5]. Although moisturizers and topical steroid therapy are commonly used for the treatment of AD, their ineffectiveness and side effects limit their use. Therefore, recent advancements in new treatment methods using herbal medicines with similar efficiency and reduced side effects are in progress [6].
The mushroom Cordyceps militaris (CM), an obligatory parasite that grows on insects or insect larvae, belongs to the family Clavicipitaceae and class Ascomycetes. CM, which is also an inhibitor of macrophage activation, has broad medical applications due to its varied pharmacological activities, including antioxidant, hypoglycemic, hypolipidemic, anti-inflammatory, antitumor, antibacterial, and antifungal activities [7]. Furthermore, it contains bioactive compounds, such as cordycepin (30-deoxyadenosine) and 20-deoxycoformycin [8-10]. CM extracts have demonstrated antioxidant, anti-inflammatory, and protective activities against acute hepatotoxicity in a murine macrophage model and in rats [11]. However, their activity on AD has not been demonstrated.
The present study, therefore, was designed to identify the nutraceutical composition of aqueous CM extracts and their anti-atopic dermatitis properties on tumor necrosis factor-α/interferon-γ (TNFα/IFNγ)-stimulated HaCaT cells and Dermatophagoides farinae extract/2,4-dinitrochlorobenzene (DFE/DNCB)-induced AD in BALB/c mice.
METHODS
Cordyceps militaris
CM was obtained from the National Institute of Horticultural and Herbal Science of Korea (Eumsung, Korea). CM specimens were lyophilized and subjected to a 2 h extraction in distilled water (DW) at 100 °C. The extract was filtered through a 0.25 μm pore filter and lyophilized in a freeze dryer for 5 days. TRIzol reagent used for RNA extraction was obtained from Invitrogen (Carlsbad, CA, USA). Primary antibodies and peroxidase-conjugated secondary antibodies were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA). All other reagents were of the highest commercially available grade.
Analysis of polysaccharides and monosaccharides
The total polysaccharide content of the extract was determined using the phenol-sulfuric acid method with Dglucose as a reference [12]. For polysaccharide analysis, 1 mg of protein-bound polysaccharide was hydrolyzed in 2 M trifluoroacetic acid (TFA) followed by evaporation. A highpressure liquid chromatography (HPLC) system (Waters Corp., Milford, MA, USA) with a Sugar-Pak column (Millipore, Tokyo, Japan) and a differential refractive index (RI) detector (RID-6A) was used to detect monosaccharides at 80 °C.
Analysis of protein and amino acid content
The protein concentration of the extract was determined using the Bradford method [13]. Bovine serum albumin (BSA) (0.1-1.0 mg mL-1) was used to construct a standard calibration curve. For the analysis of amino acids, the extract was hydrolyzed in 6 N hydrochloric acid in vacuum-sealed tubes at 110 °C for 24 h. The amino acid content of the extract was determined using an amino acid analyzer (L-8900, Hitachi High-Technologies Corp., Tokyo, Japan) with postcolumn derivatization with ninhydrin. The amino acid analyzer conditions were as follows: ion exchange column (4.6 × 60 mm), injection volume of 20 μL, UV detector, VIS1: 570 nm, and VIS2: 440 nm. An amino acid standard solution (016-08641, Wako, Osaka, Japan) was used for identification and quantification of amino acid content.
Phytochemical content
The total polyphenolic (TP) content of the extract was determined using a recently developed Fast Blue BB (FBBB) method [14]. First, the aqueous extract was diluted with deionized water (DW). The TP analysis consisted of adding 0.1 mL of 0.1% FBBB diazonium dye to 1 mL of the diluted sample, followed by the addition of 0.1 mL of 5% NaOH. After a 90-min reaction time, the absorbance was measured at 420 nm using a UV/Vis spectrophotometer (Shimadzu UV-1650 PC, Shimadzu, Tokyo, Japan). The standard curve was generated using 15 - 250 μg mL-1 gallic acid. The TP content was expressed as gallic acid equivalents (GAE) per g extract in DW.
The flavonoid content of the extract was determined using a colorimetric method described by Jia et al. [15] with modifications introduced by Barros et al. [16]. The extract (250 μL) was mixed with 1.25 mL of Milli-Q water (MQ) and 75 μL of 5% NaNO2 solution. After 5 min, 150 μL of 10% AlCl3 solution was added, and 6 min later, 500 μL of 1 M NaOH and 275 μL of MQ were added to the mixture, which was then thoroughly mixed. The intensity of the pink color of the solution was measured at 510 nm using a UV/Vis spectrophotometer (Shimadzu UV-1650 PC, Shimadzu, Tokyo, Japan). (+)-Catechin was used to generate the standard curve (0.022 - 0.34 mM). The flavonoid content was expressed as mg of (+)-catechin equivalents (CEs) per g of extract.
The β-carotene and lycopene contents of the extract were determined according to a method used in a previous study [17]. The dried extract (100 mg) was vigorously shaken with 10 mL of an acetone-hexane mixture (4:6) for 1 min and filtered through Whatman No. 4 filter paper. The absorbance of the filtrate was measured at 453, 505, and 663 nm. The amounts of β-carotene and lycopene were calculated using the following equations:
Lycopene (mg per 100 mL) = -0.0458A663 + 0.372A505 − 0.0806A453
β-carotene (mg per 100 mL) = 0.216A663 − 0.304A505 + 0.452A453
The results of these analyses were expressed as μg of carotenoid per g-1 of extract.
Fourier-transform infrared (FT-IR) spectroscopy
FT-IR spectra were obtained using an Agilent Cary 630 spectrophotometer (Agilent Technologies, Wilmington, DE, USA) equipped with a single bounce diamond ATR module for potassium bromide (KBr) and the MicroLab and Resolutions Pro Software. FT-IR spectra were obtained in transmission mode in the 350-6,300 cm−1 range, at a resolution of 2 cm−1.
Cell culture
HaCaT cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum and 10 μg/100 mL penicillin/streptomycin in a 5% CO2 atmosphere at 37 °C. For the migration assay, HaCaT cells were stimulated with TNFα (10 ng/mL) and INFγ (10 ng/mL). After 6 h of stimulation, cells were harvested, and total RNA and protein were isolated.
Animals
Eight week-old female BALB/c mice were purchased from Samtako BioKorea Co., Ltd. (Osan, Republic of Korea) and housed under specific pathogen-free conditions. All experiments were approved by the Institutional Animal Care and Use Committee of Konkuk University (Case #KU14011).
Induction of AD lesions in the ear
AD was induced in the mice by repeated local DFE (house dust mite extract) and DNCB exposure on the ears, as previously described [18]. For AD induction, the mice were divided into four groups (control, AD-only, AD + CM extract: 250 mg/kg/day, and AD + CM extract: 500 mg/kg/day). The surfaces of both earlobes were stripped five times using surgical tape (Nichiban, Tokyo, Japan). After stripping, 20 μl of 1% DNCB was painted on each ear, followed 4 days later with 20 μl of DFE (10 mg/mL). DNCB or DFE treatment was administered alternately once per week for 4 weeks. The animals received the CM extract (250 or 500 mg/kg/day orally) throughout the 4 weeks of AD induction.
Ear thickness was measured 24 h after DNCB or DFE application by using a dial thickness gauge (ID-C1012XBS, Mitutoyo Corp., Kawasaki, Japan). At days 14 and 28, blood samples were collected using orbital puncture. Plasma samples were prepared from the blood samples and stored at −70 °C for further analysis. After blood collection, the ears were removed and used for histopathological analysis. Serum immunoglobulin (IgE) and IgG2a levels were measured at days 14 and 28 after the first induction by using an IgE enzyme-linked immunosorbent assay kit (Bethyl Laboratories, Inc., Montgomery, TX, USA) according to the manufacturer’s instructions.
Histological observations
The excised ears were fixed in 4% paraformaldehyde for 16 h and embedded in paraffin. Subsequently, thin (6 μm) ear sections were stained with hematoxylin and eosin (H&E), and the epidermal and dermal thickness were measured under a microscope. Microphotographic images were obtained at a magnification of 100 ×. To measure mast cell infiltration, skin sections were stained with toluidine blue, after which the number of mast cells was counted in five randomly chosen fields of view.
Analysis of mRNA expression
For the reverse transcription polymerase chain reaction (RT-PCR), the total cellular RNA was isolated from the ear tissue of the mice in each treatment group using TRIzol reagent according to the manufacturer's protocol [18]. The firststrand complementary DNA (cDNA) was synthesized using Superscript II reverse transcriptase (Invitrogen). The conditions used for RT-PCR were similar to those previously described in related studies [18]. Quantitative PCR (qPCR) was performed in triplicate by using 12.5 μL of SYBR Premix Ex Taq (Takara Bio Inc., Shiga, Japan) and 2 μL of cDNA as a template in a final volume of 25 μL. Prior to being subjected to 40 cycles of amplification, the mixture was incubated for 15 min at 95 °C. The denaturation was performed for 30 s at 95 °C, annealing was performed in a transitional temperature range from 58 °C to 62 °C, with an increase of 0.5 °C per cycle, and the extension was performed for 30 s at 72 °C with fluorescence detection at 72 °C after each cycle. After the final cycle, melting-point analyses of all the samples were performed within a temperature range from 65 °C to 95 °C, with continuous fluorescence detection. Target gene mRNA levels were normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) levels using the following formula:
Relative mRNA expression = 2− (Ct of target gene − Ct of GAPDH),
where Ct is the threshold cycle value. In each sample, the expression level of the analyzed gene was normalized to that of GAPDH and presented as a relative mRNA level.
Statistical analysis
Statistical analysis was carried out using SAS statistical software (Release 9.1.3, SAS Institute, Cary, NC, USA). Multiple group data were analyzed using one-way analysis of variance (ANOVA) followed by Dunnett’s multiple range test. All results were expressed as the mean ± standard deviation of comparative fold differences. Data are representative of three independent experiments. The threshold for statistical significance was set at P<0.05.
RESULTS
Partial chemical characterization of CM
Confirmatory results of the nutritional value of the CM extract are shown in Table 1. The total carbohydrate and monosaccharide composition of the CM aqueous extract was determined in our series of experiments. The aqueous CM extract contained 419.21 ± 1.91 μg/g of total carbohydrate. The monosaccharide constituents of the extract were primarily glucose (264.01 ± 3.22 μg/mL) and mannose (6.36 ± 1.29 μg/mL) (Table 1). Furthermore, our results demonstrated that the CM aqueous extract was rich in amino acids. It contained 338.92 ± 1.91 μg/g of total protein with the primary amino acid being glutamic acid (3.1316 g/100 g) as shown in Table 1. The results of the phytochemical screening are provided in Table 1. Polyphenols were the primary constituents of the extract (19.79 ± 1.02 mg/g), followed by flavonoids (275.52 ± 11.01 μg/g). The contents of β-carotene and lycopene were 24.51 ± 0.25 μg/g and 3.42 ± 0.09 μg/g, respectively.
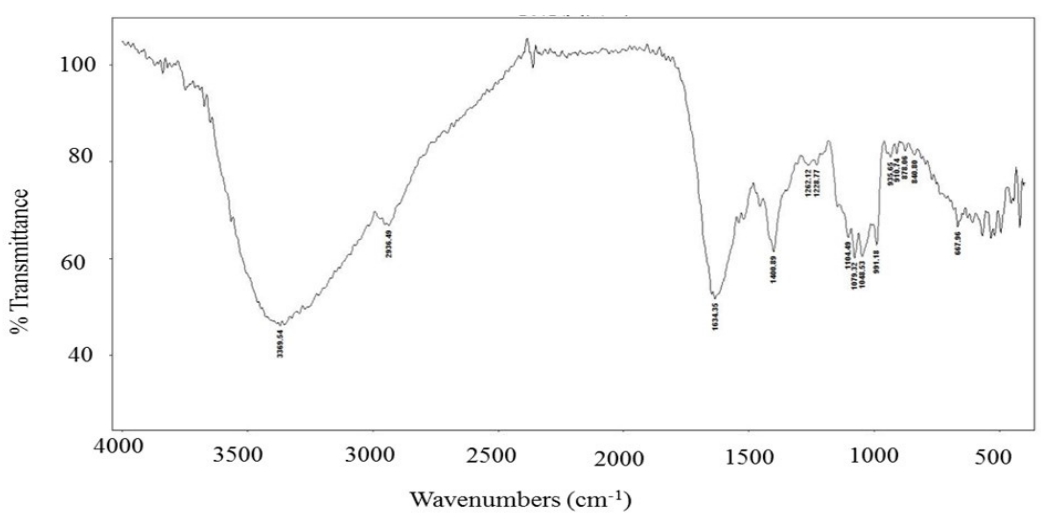
The FT-IR spectrum of the CM extract is shown in Figure 1. The bands at 3369 cm−1 indicate hydroxyl stretching in hydrogen bonds and N-H vibrations. The band at 2936 cm−1 was attributed to the C-H stretching vibration, whereas the band at 1634 cm−1 indicated asymmetric vibration of the carboxylic group. The peak at 1400 cm−1 was associated with the typical stretching frequencies of the OH groups of phenolic compounds. Peaks at 1104, 1079, and 1048 cm−1 were associated with the C-O-C linkages of sugars remaining in the extract [25]. Our data are consistent with those of other researchers who reported that the bands at 3,405 and 2,930 cm-1 were attributed to OH and C-H bond stretching, respectively, while bands at 1,420 and 1,366 cm-1 arose from the bending modes of CH2, CH, and OH [26]. Intense overlapping bands in the region of 800-1300 cm-1 correspond to CO, CC stretching, and COH bending modes.
Inhibitory effect of CM on inflammatory stress in stimulated keratinocytes
The ability of CM to block the effects of the pro-inflammatory mediators TNFα (10 ng/mL) and IFNγ (10 ng/mL) in HaCaT keratinocytes was assessed by using RT-PCR (Figure 2A). The CM extract reduced the mRNA expression levels of TNFα, chemokine C-C motif ligand 17 (CCL17), interleukin-1β (IL-1β), and interleukin-6 (IL-6) following TNFα/IFNγ-induction in a dose-dependent (0.25, 0.5, and 1.0 mg/mL) manner. This indicated that the CM extract could inhibit chemoattractant and pro-inflammatory cytokine production.
Intracellular signaling induced by CM was investigated by examining the phosphorylation of extracellular signalregulated kinases 1/2 (ERK1/2), c-Jun NH2-terminal kinases (JNKs), and p38 in TNFα/IFNγ-induced HaCaT cells. The expression levels of phosphorylated ERK1/2 and p38 were increased in cells treated with TNFα/IFNγ. Furthermore, pretreatment with CM extract suppressed the phosphorylation of ERK1/2 and p38 in TNFα/IFNγ-stimulated HaCaT cells (Figures 2B, 2C). However, neither the CM extract nor the positive control suppressed JNK activation (Figure 2D), suggesting that the CM extract only blocked the activation of ERK1/2 and p38 MAP kinase. Moreover, the inhibition of AD by the CM extract was mediated through inhibition of the overexpression of pro-inflammatory cytokine-related mRNA associated with p38 and ERK1/2 but not with JNK activation, in HaCaT cells.
CM reduced AD symptoms in a murine model
After orally administering the CM extract to mice, ear thickness was examined (Figure 3). While the ear thickness and AD lesions significantly increased following the application of DFE and DNCB, the CM extract led to a reduction in DFE/DNCB-induced ear thickness (Figure 3A, 3B). Thickening of the epidermis and dermis was also observed using a microscope, and a significantly reduced epidermal and dermal thickness was observed in a dose-dependent manner in the CM extract-treated group than in the AD control group (Figures 3C, 3D).
Allergic responses associated with mast cells originate in myeloid stem cells. Therefore, a histological analysis of atopic ears was performed to examine its correlation with the visual evaluation of AD symptoms. Excised ears from each group were stained with toluidine blue to identify infiltration by mast cells. Compared with the AD control group, the CM extract-treated group showed a significantly reduced number of infiltrated immune cells, such as mast cells (P<0.05) (Figures 4A, 4B).
The CM extract-treated mice had significantly reduced total IgE and IgG2a levels in comparison with those of the DFE/DNCB-treated mice (P<0.05) (Figure 4C, 4D). These data suggest that the potential therapeutic effect of the CM extract in AD progression is related to the downregulation of serum Ig levels.
To further explain the fundamental mechanism underlying the effects of the CM extract, the mRNA expression levels of AD-related pathogenic cytokines were examined in ear tissues by using qPCR. All cytokine-related mRNAs that were tested were upregulated in ear tissues from the AD group. The CM extract significantly reduced Th2 cytokines and Th1-related cytokine TNFα in ear tissue in a dosedependent manner (Figure 5).
DISCUSSION
Edible mushrooms are an attractive source of many different nutraceuticals, such as polysaccharides, polyphenols, peptides, lycopenes, and carotenoids [19]. CM extracts are a rich source of proteins, carbohydrates, and selected phytochemicals. A previously published report identified four different exopolysaccharides with molecular masses ranging from 50 kDa to 2260 kDa from a newly isolated Cordyceps species, Cordyceps militaris NG3 [20]. Furthermore, another research group isolated four polysaccharides from CM [21], which interrupted the progression of hepatocellular carcinoma while also demonstrating anti-inflammatory properties in rodents [22]. Therefore, considering the pharmacological activity of CM, the determination of the carbohydrate content of the CM extract is a meaningful scientific advancement. The identified polyphenolic compounds include several different subclasses including flavonoids and phenolic acids that display a large diversity of structures and functions. Mushroom fruiting bodies are reportedly rich in polyphenols and carotenoids, and possess a variety of pharmacological activities, such as antioxidant and anti-inflammatory activities, in biological systems [23]. To further our understanding of the pharmacological activity of CM and its polyphenol and flavonoid content, the main compounds contributing to the antioxidant properties of mushrooms as well as the levels of two carotenoids (β-carotene and lycopene) were determined. Our data are consistent with those of another study that also determined the polyphenol, β-carotene, and lycopene content of dried, edible mushrooms [24].
The biological effects of cytokines on keratinocytes demonstrate the fact that keratinocytes are direct targets of specific cytokines involved in skin inflammation [27]. Furthermore, pro-inflammatory cytokines, such as the interleukin (IL) family cytokines, are important regulators of epidermal functions, as their receptors have been detected in keratinocytes [28]. In addition, IFNγ is associated with the maintenance of chronic AD by increasing the expression of chemoattractants CCL17 (TARC) and CCL22 (MDC), which are related to the production of effector T cells at the inflamed site [29]. The CM extract reduced TNFα, CCL17, IL-1β, and IL-6 mRNA expression against TNFα/IFNγ-induction, suggesting that it inhibits chemoattractant and pro-inflammatory cytokine production.
Specific intracellular processes are involved in the recognition and response to extracellular stimuli, such as the signaling cascade resulting in activation of mitogen-activated protein kinases (MAPKs). The most widely studied groups of MAPKs are the ERK1/2, JNKs, and p38 kinases, which play a significant role in chronic inflammatory disease processes and skin homeostasis [30]. In the present study, the CM extract only blocked the activation of ERK1/2 and p38 MAP kinase but did not affect the activation of JNK in HaCaT cells.
In order to evaluate the immunomodulatory activity of the CM extract in vivo, the extract was orally administered to AD-induced mice. Experimental AD was induced on both earlobes of BALB/c mice by alternative weekly painting of DNCB and mite extract for 4 weeks. DFE and DNCB application substantially increased ear thickness and the appearance of AD lesions; however, the CM extract-treated group showed significantly reduced epidermal and dermal thickness compared to the AD control group.
AD is associated with elevated serum IgE levels and sensitization to a variety of environmental allergens [31]. The cross-linking of IgE and the allergen activates mast cells at the sites of allergic reactions, which leads to the release of chemical mediators, including histamine and cytokines [32]. Furthermore, AD is associated with the typical Th2 type immune response, which shows elevated total IgE serum levels and Th2 type cytokine expression [33]. Elevated total IgE serum levels are a strong risk factor for AD in children from birth to six years of age, and are associated with persistent eczema in adults [34]. In contrast, higher total IgG levels have been linked with the Th1 response [25]. To determine whether the CM extract exerts its effects primarily via the Th1 or Th2 response, the serum levels of IgE (total and DFEspecific) and IgG2a were measured in each group following treatment completion. The results of this study suggest that the therapeutic effects of the CM extract in AD are related to the downregulation of serum Ig levels.
Previous reports studying the beneficial effects of the Cordyceps species in various inflammatory diseases include studies of ethanolic extracts of cultured fruiting bodies and mycelia of CM, which exhibited anti-inflammatory activity in the croton oil-induced ear edema test in mice [35]. Other studies using treatment with water extract of C. sinensis reported down-regulation of inflammation-related genes in the rat kidney [36]. Finally, a CM hot water extract inhibited lipopolysaccharide (LPS)-stimulated nitric oxide (NO) production, and TNFα and IL-6 release in RAW 264.7 cells [37]. Our results are consistent with these previous reports. Furthermore, mushroom extracts suppressed the overexpression of Th2 and Th1-related cytokines [38]. Thus, our results demonstrating that CM extract inhibits the expression of Th1 and Th2 cytokines are consistent with those of previous reports. These data support the therapeutic use of CM extract when treating both the acute and chronic stages of AD.
This study identified the nutraceutical composition and therapeutic effects of the aqueous extract of CM against AD in vitro and in vivo. CM extract inhibited TNF-α, CCL17, IL-1β, and IL-6 mRNA production and reduced ERK1/2 and p38 expression in HaCaT keratinocytes. Oral administration of the CM extract decreased the severity of AD, including histopathological signs, and significantly reduced the production of Ig and expression of pathogenic cytokines in DFE/DNCB-induced BALB/c mice. Therefore, our results indicate that the aqueous extract of CM could be utilized as an effective complementary alternative medicine to prevent or treat atopic dermatitis symptoms.