 |
 |
- Search
| Phys Act Nutr > Volume 26(2); 2022 > Article |
|
Abstract
[Purpose]
Aging and obesity are associated with skeletal muscle atrophy-related signaling pathways, including apoptosis. Many studies have shown that menopause is associated with an increased risk of skeletal muscle atrophy. There is an increasing need to develop strategies that will improve the risk of skeletal muscle atrophy through exercise interventions. However, the effect of exercise on estrogen deficiency-induced apoptosis in skeletal muscles is poorly understood. Therefore, we examined the effects of low-intensity exercise on ovariectomy (OVX)-induced apoptosis of the soleus and plantaris muscles.
[Methods]
The ovaries of all female Sprague-Dawley rats aged 8 weeks, were surgically removed to induce postmenopausal status. The rats were randomly divided into three treatment groups: (1) NSV (normal-diet-sedentary-OVX); (2) HSV (high-fat-diet-sedentary-OVX); and (3) HEV (high-fat-diet-exercise-OVX). The exercise groups were regularly running for 30-40 min/day at 15-18 m/minute, five times/week, for eight weeks.
[Results]
The mRNA levels of Bax significantly decreased in the exercised soleus muscle, and caspase-3 decreased in the plantaris. The skeletal muscle TUNEL-positive apoptotic cells in the high-fat-diet-sedentary OVX rats improved in the treadmill exercise group. Additionally, nuclear caspase-3 levels decreased in the treadmill exercise group compared to those in both sedentary groups. These results suggest that low-intensity treadmill exercise prevents skeletal muscle apoptosis in HFD-fed OVX rats.
[Conclusion]
Induction of HFD in estrogen-deficient mice increased apoptosis in skeletal muscle, which could also be alleviated by low-intensity aerobic exercise. These results may indicate a crucial therapeutic effect of treadmill exercise in preventing skeletal muscle apoptosis in menopausal or post-menopausal women.
Menopause is associated with a decline in the level of circulating estrogen, which causes an increase in metabolic diseases, such as obesity, type 2 diabetes, and cardiovascular disease [1-3]. Although numerous factors contribute to the deterioration of physiological function during menopause, an increase in fat mass is the most serious risk factor for physical dysfunction [1,3,4]. The accumulation of body fat is related to skeletal muscle inflammation, mitochondrial dysfunction, and DNA damage, which may contribute to muscle weakness and sarcopenia [5,6]. There is an increasing need to develop approaches for improving metabolic disorders and skeletal muscle wasting through nutrients, dietary supplements, hormone replacement therapy, and physical activity intervention in menopausal and post-menopausal women [3,7].
Many previous studies have reported that ovariectomy (OVX) animal models show an increase in body weight, inflammation, insulin resistance, and fat accumulation in abdominal, liver, heart, and skeletal muscles [8-11]. However, several studies have suggested that OVX-induced metabolic abnormalities can be improved by endurance treadmill training exercise for 8 or 12 weeks [9,11]. Ovariectomy is also known to induce a decline in skeletal muscle mass, myosin dysfunction, and muscle contractile proteins [12-14]. Interestingly, OVX-induced muscle impairment was prevented by voluntary wheel running in a rat model [12,15]. Additionally, 10 weeks of treadmill exercise suppressed OVX-induced cardiac apoptosis in a rat model [2]. However, given that OVX animals are an applicable model for postmenopausal women, it is necessary to consider high fat accumulation in postmenopausal women and to reflect on the experimental design that low-intensity exercise is a viable form of exercise intervention. These prior designs and results prompted us to investigate whether regular bouts of low-intensity treadmill exercise played a role in regulating skeletal muscle apoptosis in high-fat-induced OVX rat skeletal muscles. Additionally, based on the fact that the soleus and plantaris muscles of rats contain high levels of slow (type I) and fast (type 2b) fibers, respectively [16], we examined whether there is a difference in apoptosis by muscle fiber type.
Apoptosis is an important mechanism in skeletal muscle mass. Previous studies have suggested that an increase in body fat alters signaling pathways and induces apoptosis in human and animal models [6,17,18]. Sishi et al. reported that high-fatdiet (HFD)-induced obesity increases apoptosis-related tumor necrosis factor-ɑ (TNF-ɑ) receptor expression and caspase-3 activity in the gastrocnemius muscle of a pre-diabetic rat model [6]. These studies suggest a possible interaction between estrogen deficiency-induced obesity and skeletal muscle apoptosis. Additionally, Fonseca et al. reported that physical inactivity is a major contributor to menopause induced muscle atrophy. However, the effects of exercise on the modulation of apoptosis have not yet been demonstrated in HFD-induced obese OVX rat skeletal muscles [12].
This study aimed to determine whether eight weeks of low-intensity treadmill exercise modulates apoptosis in highfat-fed OVX soleus and plantaris muscles. We hypothesized that a HFD and ovariectomy would increase the expression of pro-apoptotic genes (Bax and caspase-3). Additionally, the number of terminal deoxynucleotidyl transferase dUTP-mediated nick-end labeling (TUNEL)-positive cells increased. However, eight weeks of treadmill exercise may help mitigate the HFD-fed and OVX-induced pro-apoptotic phenomena in the soleus and plantaris muscles. The study also aimed to evaluate fiber type-specific responses to the anti-apoptotic effect of treadmill exercise in OVX rat skeletal muscles.
All animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Ewha Womans University, Seoul, Korea. Permit Number: 14-038.
Female Sprague-Dawley rats (8 weeks old) were obtained from the Central Lab Animal (Seoul, Korea). The animals were housed in an air-conditioned room at 23 ± 1°C and 64.1% relative humidity with a light/dark cycle of 12 h. The ovaries of all the rats were surgically removed to induce postmenopausal status, and one week of recovery period was provided. After the recovery period, the rats were randomly divided into three treatment groups: (1) NSV, (2) HSV, (3) HEV. The HFD were prepared daily for eight weeks using premade diets. The HFD contained 45% energy as fat derived from corn oil and lard, and a normal diet containing 10% energy as fat (Research Diet Inc., New Brunswick, NJ).
After one week of ovariectomy surgical recovery period, the animals were acclimatized to running on a treadmill for 15 min at 8 m/min at 0° inclinations on the first day. Subsequently, the animals were regularly trained five days/week, for eight weeks, and the training started at 10 am. From one to four weeks, the animals ran on a treadmill for 30 min at 15 m/min with 0° inclinations. Between 5-8 weeks, the training subsequently progressed to 40 min and 18 m/min with 0° inclinations. Okamoto et al. reported that an exercise intensity below the lactate threshold did not stress rats, and an exercise intensity of ~20 m/min was the lactate threshold in rats '[19]. The exercise training protocol used a modified version of previous studies [20,21]. All the rats were restrained from training 24 h before sacrifice.
The rats were exposed to CO2, and the soleus and plantaris muscles were freshly dissected, trimmed, dried with filter paper, and weighed using an electronic balance scale (OHAUS, Parsippany, NJ, USA). The muscles were snap-frozen in liquid nitrogen for RNA extraction. To prepare paraffin sections, the muscles were preserved in formalin solution, containing 4% paraformaldehyde for three days at 4°C (Sigma, St. Louis, MO).
Total RNA was extracted from the soleus and plantaris muscles using TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA). The RNA concentration and quality were measured at 260/280 nm using a spectrophotometer (Nanodrop-2000, Thermo Fisher Scientific, Waltham, MA). Subsequently, cDNA was synthesized from 1 μg of total RNA in the presence of a random primer, 2.5 mM dNTP, RNase inhibitor and reverse transcriptase (Invitrogen Life Technologies) in a final volume of 20 μg at 25°C for 10 min, followed by 42°C for 60 min, and 95°C for 5 min. Real-time quantitative polymerase chain reaction (qPCR) was performed using the Step-One-Plus system (Applied Biosystems, Foster City, CA). The qPCR was performed using SYBR Green Master Mix (Bioline, London, UK), according to the manufacturer’s instructions. Primer sets for the target genes are listed in Table 1. The primers were purchased from Macrogen (Macrogen Inc., Seoul, KOREA). Expression of target genes was normalized to that of Glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and the relative expression of all genes was calculated using comparative cycle threshold (CT) method.
Terminal TUNEL assay was conducted using the ClickiT Plus TUNEL assay kit (Thermo scientific), according to the manufacturer’s instructions. Paraffin-embedded sections were deparaffinized and hydrated, and antigen retrieval was performed using xylene. Tissues were permeabilized with proteinase K for 15 min at room temperature (RT) Subsequently, the slides were washed twice with phosphate buffered saline (PBS) for 5 min, after which they were reacted with the EdUTP nucleotide mixture, terminal deoxynucleotidyl transferase (TdT) enzyme, and TdT reaction mixture for 60 min at 37°C. The slides were then washed with 3% bovine serum albumin (BSA) in 0.1% Triton X-100 in PBS, after which they were incubated with a TUNEL reaction cocktail, including alexa fluor-488 dyes for 30 min at 37°C. Subsequently, the slides were washed with 3% BSA in PBS, after which they were stained with 4’,6-diamidino-2 phenylindole (DAPI) at a concentration of 1.5 μg/mL to localize the nuclei. Finally, the slides were mounted and photographed using a Nikon imaging system (Nikion).
The soleus and plantaris muscles were placed in formalin solution containing 4% paraformaldehyde (Sigma-Aldrich). Cross-sections were cut from the midbelly region of each muscle. Formalin-fixed paraffin-embedded sections (4-5 μm) were deparaffinized and hydrated, and antigen retrieval was performed using xylene. The tissue was made permeable with 0.02% Triton X-100 in PBS (PBST) for 15 min and blocked with 5% BSA in PBST for 30 min. Subsequently, the slides were washed with PBS once and then probed with caspase-3 polyclonal rabbit antibody (Cell Signaling, Beverley, MA) at a dilution ratio of 1:500 overnight at 4°C in the PBS containing 5% BSA. The slides were then washed thrice for 5 min each in 0.05% Tween 20 in PBS, after which they were incubated with Alexa 568-conjugated goat anti-rabbit IgG secondary antibody (Invitrogen Life Technologies) diluted at 1:200 for 20 min at room temperature in PBS containing 5% BSA. Subsequently, the slides were washed three times with 0.05% Tween 20 in PBS, after which they were stained with DAPI at a concentration of 1.5 μg/mL to localize the nuclei. Finally, the slides were mounted and photographed using a Nikon imaging system (Nikon, Tokyo, JAPAN).
To test whether low-intensity treadmill exercise is associated with the expression of apoptosis-related genes, mRNA levels were determined using qRT PCR. The expression of genes involved in pro-apoptosis (Bax and caspase-3) was measured in the soleus and plantaris muscles. The GAPDH mRNA expression levels were used as a correction factor. The level of caspase-3 in the soleus muscles reduced in the HEV group when compared with the HSV group (not significant) (Figure 1A). In the plantaris muscle, the caspase-3 mRNA levels were significantly elevated by the HFD compared with the NSV group (p<0.01). However, training exercise (p<0.01) significantly decreased the caspase-3 mRNA levels (Figure 1B). As shown in Figure 1C, the level of Bax mRNA was significantly elevated by the HFD when compared with the NSV group (p<0.05). However, EX significantly decreased (p<0.05) the soleus muscles when compared with the HSV group. There was no significant difference in the plantaris muscle between any pair of groups (Figure 1D).
To clarify the apoptotic activity in the HFD and OVX-induced soleus and plantaris muscles after the training exercise, a TUNEL assay was conducted. The number of TUNEL-positive (green) and DAPI-stained nuclei (blue) was determined, and the percentage of apoptotic cells was calculated as the ratio of TUNEL-positive cells relative to DAPI-stained nuclei from each muscle. As shown in Figure 2A, the number of TUNEL-positive cells in the soleus muscle of the HSV group was higher than that in the NSV group (p<0.01). However, the number of TUNEL-positive cells in the HEV group (p<0.05) was lower than that in the HSV group. We also observed that the plantaris muscle of the HSV group had a greater number of TUNEL-positive cells than that of the NSV group (p=0.000). However, the number of TUNEL-positive cells in the HEV group (p<0.01) was significantly lower than in the HSV group (Figure 2C-D). These results suggest that treadmill training exercise has an anti-apoptotic function in the OVX rat skeletal muscles.
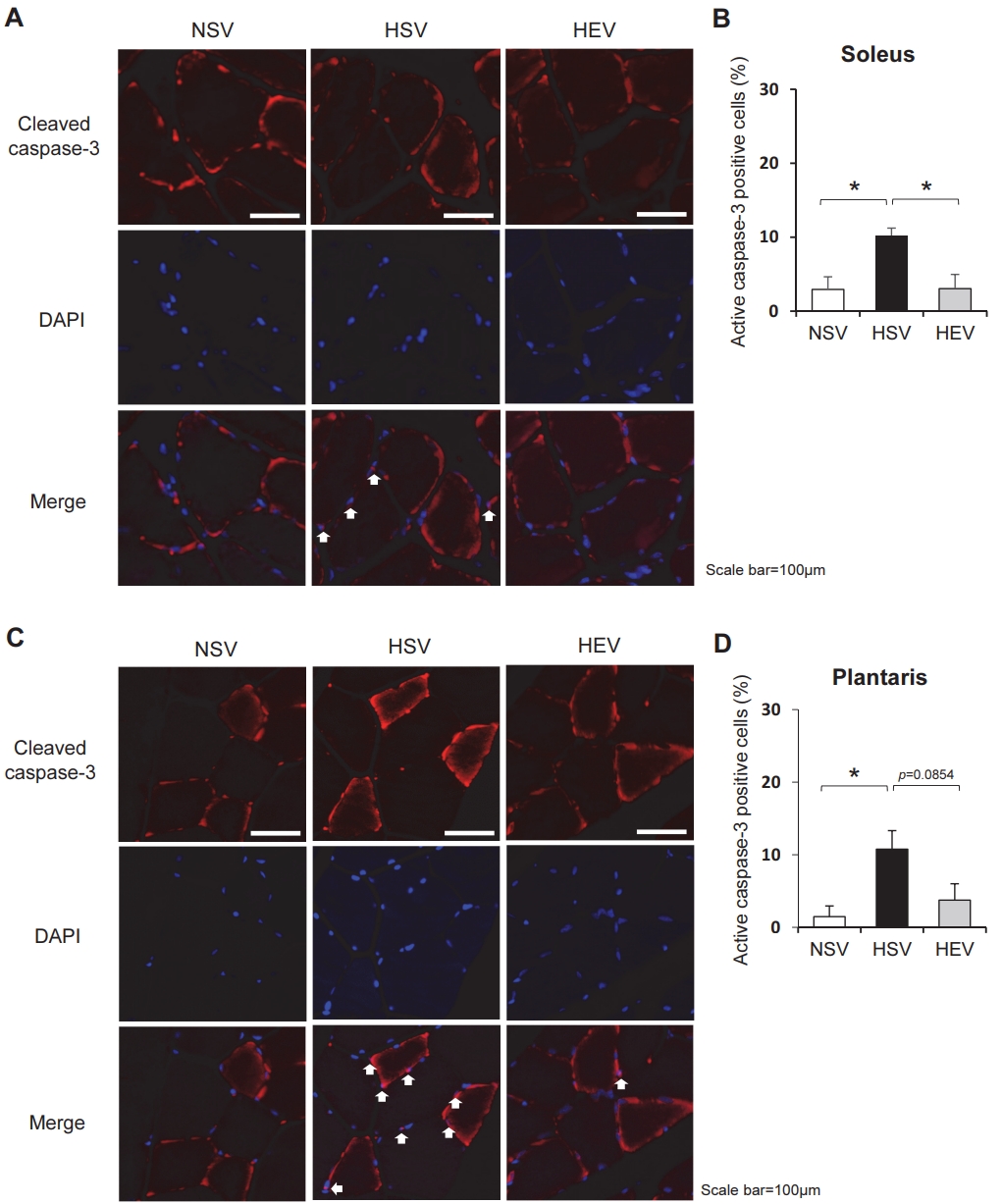
As shown in Figure 3, active caspase-3 (red) was primarily present in the cytosolic compartment in the normal-diet-sedentary group in the soleus (Figure 3A) and plantaris muscles (Figure 3B). However, a HFD led to marked enhancement of active caspase-3 nuclear (blue) translocation in both muscles. This HFD-induced caspase-3 nuclear translocation was markedly blocked by treadmill exercise in the soleus and plantaris muscles. These results indicate that even in HFD and estrogen deficiency-induced caspase-3 nuclear localization, treadmill exercise training can considerably restrict caspase-3 activity in both types of skeletal muscle fibers.
Menopause or post-menopause not only affects metabolic problems, such as cardiovascular disease, obesity, and diabetes but also affects skeletal muscle atrophy. Although estrogen deficiency is strongly linked to obesity and skeletal muscle atrophy, the molecular mechanisms underlying the relationship between estrogen reduction-induced obesity and skeletal muscle apoptosis in OVX models have not been studied. We need to understand the interaction between regular aerobic training exercise and skeletal muscle apoptosis in an estrogen deficient and HFD-induced obesity model.
Our study provided several important findings. First, HFD significantly increased apoptotic gene levels, caspase-3 activity, and TUNEL-positive apoptotic cell numbers in the OVX rat skeletal muscles. This finding implies that skeletal muscle cell apoptosis may be influenced by increased body fat mass in postmenopausal women. Second, eight weeks of treadmill exercise training significantly decreased the HFD and ovariectomy-induced apoptotic gene levels and caspase-3 activity, which decreased TUNEL-positive apoptotic cell numbers in the soleus and plantaris muscles. Low-intensity aerobic training exercise, such as walking and jogging, has been shown to be a beneficial exercise regimen for middle-aged women following menopause, with positive effects on body composition alterations [22]. Therefore, our research, which showed that low-intensity aerobic exercise can reduce skeletal muscle apoptosis, is regarded as a useful study that can be applied in postmenopausal women.
Many previous studies have shown that long-term highfat feeding in a mouse model, leptin receptor-deficient db/db mice, and obese Zucker rat models lead to muscle protein degradation and apoptosis [6,23]. Additionally, estrogen deficiency-induced fat accumulation promotes decreases in muscle strength, cross-sectional area, satellite cell activity, and mitochondrial function in human and animal models [9,24-26]. These results strongly suggest that estrogen deficiency interferes with skeletal muscle plasticity and is partly responsible for fat accumulation in OVX animal models and post-menopausal women. However, the exact mechanisms underlying the loss of skeletal muscle mass following estrogen deficiency-induced obesity are largely unknown.
Generally, estrogen deficiency-induced obesity and type 2 diabetes are associated with physical inactivity, lower energy expenditure, and elevated levels of pro-inflammatory cytokines, such as tumor necrosis factor-α (TNF-ɑ) [4,6,25]. These factors lead to a decrease in the activity of muscle function and energy metabolism-related signaling pathways, such as the mammalian target of rapamycin (mTOR)-p70S6K pathway and AMP-activated protein kinase (AMPK) pathways [27-29] that signaling cascade changes would result in increased forkhead box transcription factors (FOXO)-dependent transcription of muscle-specific E3 ubiquitin ligases, atrogin-1/MAFbx, and MuRF-1 [6,27]. In our previous study, HFD-induced obesity resulted in a significant decrease in AMPK activity and an increase in E3 ubiquitin ligase protein expression, but training exercise had anti-atrophy effects in an OVX rat model [30]. Liang et al. reported that sustained aerobic exercise prevented muscle protein degradation by inhibiting apoptosis through the AMPK/ proliferator-activated receptor-gamma coactivator-1α (PGC-1α) signaling pathway in aged mice [31]. It is presumed that this mechanism supports the results of our study that low-intensity aerobic exercise training suppressed the expression of apoptosis-related genes.
Several studies have shown that apoptosis in response to the activation of the major pro-apoptotic factor, Bax, enhances cytochrome c release from the mitochondria and increases the activity of caspase-3 to accelerate apoptosis through disassembly of cell structures in various cell types [6,32,33]. These pro-apoptotic markers, such as Bax, cytochrome c, caspase-9, and caspase-3 are activated and expressed after surgical ovariectomy in rat cardiac muscle. However, 10 weeks of treadmill training exercise attenuated the ovariectomy-induced cardiac muscle apoptosis [2]. They also conducted the TUNEL assay in cardiac muscles from the left ventricles. Consequently, the number of TUNEL-positive cells in the OVX exercise-trained group was lower than that in the OVX non-exercise group. Our experiment showed the same tendency for the soleus and plantaris muscles. Although our study design was rather a severe metabolic condition, low-intensity treadmill training exercise effectively prevented pro-apoptotic gene levels and decreased caspase-3 activity and TUNEL-positive cell numbers.
The expression of caspase-3 and Bax in the soleus and plantaris muscles was confirmed to be different. Apoptosis is thought to occur in the soleus muscle composed of type1 fibers with high mitochondrial content due to intrinsic regulation that increases Bax mRNA expression. In the plantaris muscle composed of type 2 fibers, apoptosis occurs by extrinsic regulation, such as Fas receptor or TNFα. Koçtürk et al. reported that the cause of apoptosis can be divided into intrinsic and extrinsic, according to the mitochondrial content of the muscle fibers [34].
We hypothesized that a HFD, characterized by rapid fat gain, will make skeletal muscles more susceptible to apoptosis in the context of ovariectomy-induced estrogen deficiency. Therefore, we fed the OVX model a HFD to manipulate the characteristics of the menopause model, which is characterized by excessive fat accumulation. Consequently, the high-fat-diet used in this study successfully induced apoptosis. A remarkable finding was that eight weeks of treadmill training exercise inhibited apoptosis in the hind limb muscles of the HFD-fed OVX rats. However, since we did not use the OVX-operated rats as a control group, we cannot explain that estrogen deficiency itself affected skeletal muscle apoptosis. This can be explained by the fact that low-intensity aerobic exercise effectively inhibited the apoptosis of skeletal muscle increased by a HFD in the estrogen deficiency model.
Regular training exercise has powerful beneficial effects on hormonal, metabolic, and skeletal muscle system [8,21,35]. It is important to note that estrogen replacement significantly attenuated OVX-induced deleterious factors in skeletal muscles. Estrogen replacement therapy prevents body mass gain, visceral fat mass, and muscle atrophy in OVX models [3,14,25]. However, many researchers also have significant concerns regarding the side effects of these estrogen replacement therapies. Regular physical activity is the most effective intervention for postmenopausal women [12,36]. Our study provides new insights into the function of low-intensity treadmill exercise and suggestions for the treatment of estrogen deficiency-induced pathological conditions, such as skeletal muscle apoptosis and atrophy.
In summary, our study suggests a role for low-intensity aerobic treadmill exercise in high fat-induced apoptosis in OVX rat skeletal muscle. We found that eight weeks of treadmill exercise significantly blocked ovariectomy and HFD-induced pro-apoptotic activity. Altogether, our data may be an important evidence that could improve the menopausal induced skeletal muscle atrophy and wasting.
Acknowledgments
This work was supported by the Basic Science Research Program of the National Research Foundation of Korea (NRF-2019R1I1A1A01061357).
Figure 1.
Apoptosis-related genes, bax and caspase-3 mRNA levels, determined by real-time PCR in OVX and HFD rat soleus and plantaris muscles at the end of an eight-week period of receiving EX. NSV, OVX + normal diet; HSV, OVX + HFD; HEV, OVX + HFD + EX. Target mRNA values are shown to normalized the GAPDH mRNA level for each sample. The values are means ± SE with n=7 for each condition. *p<0.05, and **p<0.01.
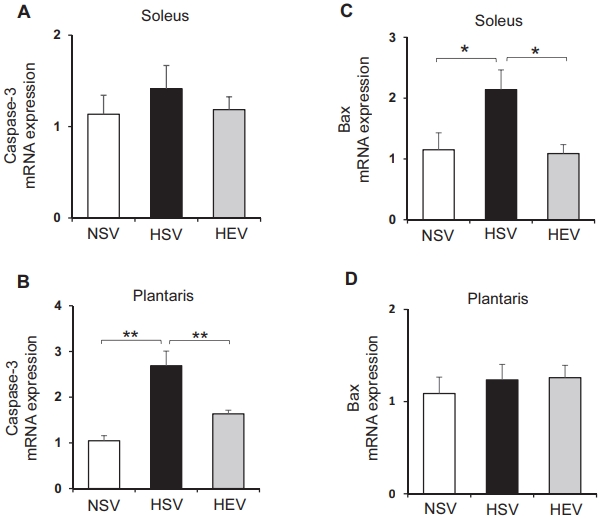
Figure 2.
TUNEL assay revealed the undergoing apoptosis in the soleus (A-B) and plantaris (C-D) muscles from high-fat-fed ovariectomized rats. Each muscle sections stained with TUNEL (green) and nuclei (blue). Apoptosis cells indicated by arrows. NSV, OVX + normal diet; HSV, OVX + HFD; HEV, OVX + HFD + EX. The quantitative analysis of TUNEL-stained apoptotic cells in each muscle. The values are means ± SE with n=3 for each condition. *p<0.05, **p<0.01, and ***p=0.000.
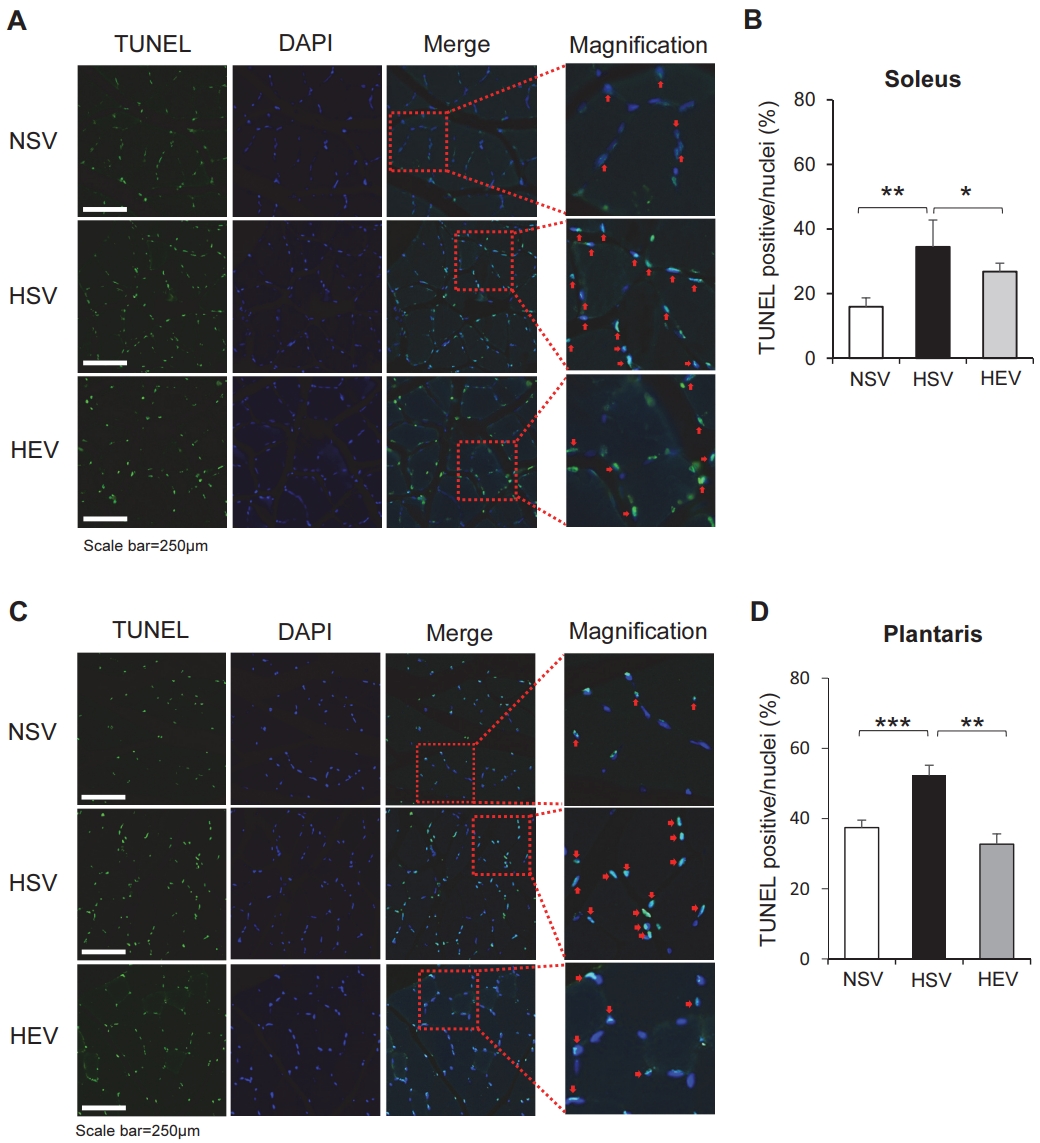
Figure 3.
Immunohistochemical staining image showing high-fat-fed and ovariectomy-induced active caspase-3 (red) translocation into the nucleus (blue) in the soleus (A-B) and plantaris (C-D) muscles. NSV, OVX + normal diet; HSV, OVX + HFD; HEV, OVX + HFD + EX. The values are means ± SE with n=3 for each condition. *p<0.05.
REFERENCES
1. Lacey JV Jr, Mink PJ, Lubin JH, Sherman ME, Troisi R, Hartge P, Schatzkin A, Schairer C. Menopausal hormone replacement therapy and risk of ovarian cancer. JAMA 2002;288:334-41.


2. Huang CY, Lin YY, Hsu CC, Cheng SM, Shyu WC, Ting H, Yang AL, Ho TJ, Lee SD. Antiapoptotic effect of exercise training on ovariectomized rat hearts. J Appl Physiol 2016;121:457-65.


3. Tiidus PM, Lowe DA, Brown M. Estrogen replacement and skeletal muscle: mechanisms and population health. J Appl Physiol 2013;115:569-78.



5. Brons C, Grunnet LG. Mechanisms in endocrinology: skeletal muscle lipotoxicity in insulin resistance and type 2 diabetes: a causal mechanism or an innocent bystander? Eur J Endocrinol 2017;176:R67-78.


6. Sishi B, Loos B, Ellis B, Smith W, du Toit EF, Engelbrecht AM. Diet-induced obesity alters signalling pathways and induces atrophy and apoptosis in skeletal muscle in a prediabetic rat model. Exp Physiol 2011;96:179-93.



7. Enns DL, Tiidus PM. The influence of estrogen on skeletal muscle: sex matters. Sports Med 2010;40:41-58.

8. Leite RD, Prestes J, Bernardes CF, Shiguemoto GE, Pereira GB, Duarte JO, Domingos MM, Baldissera V, de Andrade Perez SE. Effects of ovariectomy and resistance training on lipid content in skeletal muscle, liver, and heart; fat depots; and lipid profile. Appl Physiol Nutr Metab 2009;34:1079-86.


9. Pighon A, Gutkowska J, Jankowski M, Rabasa-Lhoret R, Lavoie JM. Exercise training in ovariectomized rats stimulates estrogenic-like effects on expression of genes involved in lipid accumulation and subclinical inflammation in liver. Metabolism 2011;60:629-39.


10. Rogers NH, Perfield JW 2nd, Strissel KJ, Obin MS, Greenberg AS. Reduced energy expenditure and increased inflammation are early events in the development of ovariectomy-induced obesity. Endocrinology 2009;150:2161-8.




11. Saengsirisuwan V, Pongseeda S, Prasannarong M, Vichaiwong K, Toskulkao C. Modulation of insulin resistance in ovariectomized rats by endurance exercise training and estrogen replacement. Metabolism 2009;58:38-47.


12. Fonseca H, Powers SK, Goncalves D, Santos A, Mota MP, Duarte JA. Physical inactivity is a major contributor to ovariectomy-induced sarcopenia. Int J Sports Med 2012;33:268-78.


13. McCormick KM, Burns KL, Piccone CM, Gosselin LE, Brazeau GA. Effects of ovariectomy and estrogen on skeletal muscle function in growing rats. J Muscle Res Cell Motil 2004;25:21-7.


14. Moran AL, Nelson SA, Landisch RM, Warren GL, Lowe DA. Estradiol replacement reverses ovariectomy-induced muscle contractile and myosin dysfunction in mature female mice. J Appl Physiol 2007;102:1387-93.


15. Kadi F, Karlsson C, Larsson B, Eriksson J, Larval M, Billig H, Jonsdottir IH. The effects of physical activity and estrogen treatment on rat fast and slow skeletal muscles following ovariectomy. J Muscle Res Cell Motil 2002;23:335-9.

16. Yasuda K, Nishikawa W, Iwanaka N, Nakamura E, Seino Y, Tsuda K, Ishihara A. Abnormality in fibre type distribution of soleus and plantaris muscles in non-obese diabetic Goto-Kakizaki rats. Clin Exp Pharmacol Physiol 2002;29:1001-8.


17. Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immunol 2011;29:415-45.


18. Thornell LE. Sarcopenic obesity: satellite cells in the aging muscle. Curr Opin Clin Nutr Metab Care 2011;14:22-7.


19. Okamoto M, Hojo Y, Inoue K, Matsui T, Kawato S, McEwen BS, Soya H. Mild exercise increases dihydrotestosterone in hippocampus providing evidence for androgenic mediation of neurogenesis. Proc Natl Acad Sci U S A 2012;109:13100-5.



20. Ogura Y, Naito H, Kakigi R, Ichinoseki-Sekine N, Kurosaka M, Yoshihara T, Akema T. Effects of ageing and endurance exercise training on alpha-actinin isoforms in rat plantaris muscle. Acta Physiol 2011;202:683-90.

21. Linden MA, Pincu Y, Martin SA, Woods JA, Baynard T. Moderate exercise training provides modest protection against adipose tissue inflammatory gene expression in response to high-fat feeding. Physiol Rep 2014;2:12071.




22. Krumm EM, Dessieux OL, Andrews P, Thompson DL. The relationship between daily steps and body composition in postmenopausal women. J Womens Health 2006;15:202-10.

23. Zhou Q, Du J, Hu Z, Walsh K, Wang XH. Evidence for adipose-muscle cross talk: opposing regulation of muscle proteolysis by adiponectin and fatty acids. Endocrinology 2007;148:5696-705.


24. Carr MC. The emergence of the metabolic syndrome with menopause. J Clin Endocrinol Metab 2003;88:2404-11.


25. Cavalcanti-de-Albuquerque JP, Salvador IC, Martins EL, Jardim-Messeder D, Werneck-de-Castro JP, Galina A, Carvalho DP. Role of estrogen on skeletal muscle mitochondrial function in ovariectomized rats: a time course study in different fiber types. J Appl Physiol 2014;116:779-89.


26. Lizcano F, Guzman G. Estrogen deficiency and the origin of obesity during menopause. Biomed Res Int 2014;2014:757461.




27. Akhmedov D, Berdeaux R. The effects of obesity on skeletal muscle regeneration. Front Physiol 2013;4:371.



28. Paturi S, Gutta AK, Kakarla SK, Katta A, Arnold EC, Wu M, Rice KM, Blough ER. Impaired overload-induced hypertrophy in obese Zucker rat slow-twitch skeletal muscle. J Appl Physiol 2010;108:7-13.


29. Sitnick M, Bodine SC, Rutledge JC. Chronic high fat feeding attenuates load-induced hypertrophy in mice. J Physiol 2009;587:5753-65.



30. Kim HJ, Lee WJ. Low-intensity aerobic exercise training: inhibition of skeletal muscle atrophy in high-fat-diet-induced ovariectomized rats. J Exerc Nutrition Biochem 2017;21:19-25.



31. Liang JL, Zhang H, Zeng ZZ, Wu LW, Zhang Y, Guo YJ, Lv J, Wang CY, Fan JJ, Chen N. Lifelong aerobic exercise alleviates sarcopenia by activating autophagy and inhibiting protein degradation via the AMPK/PGC-1 alpha signaling pathway. Metabolites 2021;11:323.



32. Kim HJ, Oh JE, Kim SW, Chun YJ, Kim MY. Ceramide induces p38 MAPK-dependent apoptosis and Bax translocation via inhibition of Akt in HL-60 cells. Cancer Lett 2008;260:88-95.


34. Kocturk S, Kayatekin BM, Resmi H, Acikgoz O, Kaynak C, Ozer E. The apoptotic response to strenuous exercise of the gastrocnemius and solues muscle fibers in rats. Eur J Appl Physiol 2008;102:515-24.



-
METRICS

-
- 1 Crossref
- Scopus
- 2,226 View
- 54 Download
- Related articles in Phys Act Nutr





